Catalog # |
Size |
Price |
|
|---|---|---|---|
| MB-003-30 | 1 ml | $796 |

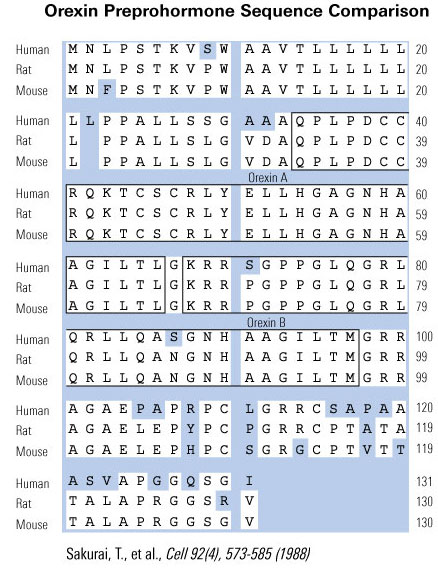

Orexin A and orexin B were initially identified as endogenous ligands for two orphan G protein-coupled receptors [104]. They were initially recognized as regulators of feeding behavior in view of their exclusive production in the lateral hypothalamic area (LHA), a region known as the feeding center, and their pharmacological activity [104,30,49,107]. Subsequently, the finding that orexin deficiency causes narcolepsy in humans and animals suggested that these hypothalamic neuropeptides play a critical role in regulating sleep/wake cycle [22,46,71,95,117]. These peptides activate waking-active monoaminergic and cholinergic neurons in the hypothalamus/brain stem regions to maintain a long, consolidated awake period. Recent studies on efferent and afferent systems of orexin neurons, and phenotypic characterization of genetically modified mice in the orexin system further suggested roles of orexin in the coordination of emotion, energy homeostasis, reward system, and arousal [3,80,106,137]. A link between the limbic system and orexin neurons might be important for increasing vigilance during emotional stimuli. Orexin neurons are also regulated by peripheral metabolic cues, including ghrelin, leptin, and glucose, suggesting that they might have important roles as a link between energy homeostasis and vigilance states [137]. Recent research has also implicated orexins in reward systems and the mechanisms of drug addiction [13,48,91]. These observations suggest that orexin neurons sense the outer and inner environment of the body, and maintain proper wakefulness of animals for survival. This review discusses the mechanism by which orexins maintain sleep/wakefulness states, and how this mechanism relates to other systems that regulate emotion, reward, and energy homeostasis.
Ohno K, Sakurai T. Orexin neuronal circuitry: role in the regulation of sleep and wakefulness. Front Neuroendocrinol. 2008;29(1):70-87
Orexins are neuropeptides that are localized in neurons within the lateral hypothalamus and regulate feeding behavior. The lateral hypothalamus plays an important role in not only feeding but also in the central regulation of gut function. Along this line, accumulating evidence has shown that orexins act in the central nervous system to regulate gastrointestinal functions. The purpose of this review is to summarize recent relevant findings on brain orexins and the digestive system, and discuss the pathophysiological roles of these peptides. Centrally administered orexin or endogenously released orexin in the brain potently stimulates gastric acid secretion in rats. The vagal cholinergic pathway is involved in the orexin-induced stimulation of acid secretion. Because of its stimulatory action on feeding, it can be hypothesized that orexin in the brain is a candidate mediator of cephalic phase gastric secretion. In addition, brain orexin may be involved in the development of depression and functional gastrointestinal disorders, which are frequently accompanied by inhibition of gut function, because lack of orexin activity might cause the inhibition of gastric physiological processes and evoke a depressive state. These lines of evidence suggest that orexin in the brain is a potential molecular target for treatment of functional gastrointestinal disorders.
Okumura T, Takakusaki K. Role of orexin in central regulation of gastrointestinal functions. J Gastroenterol. 2008;43(9):652-60
Maintenance of energy homeostasis requires the coordination of systems that regulate feeding, body temperature, autonomic and endocrine functions with those that govern an appropriate state of arousal (wakefulness). Historically, the hypothalamus has been recognized to play a critical role in maintaining energy homeostasis by integrating these factors and coordinating metabolic, neuroendocrine and behavioral responses and arousal states. Recent studies have suggested that orexin-containing neurons in the lateral hypothalamic area (LHA) constitute an important central pathway that promotes adaptive behavioral and arousal responses to metabolic and environmental signals. Orexins, also called hypocretins, are neuropeptides originally identified as endogenous ligands for two orphan G-protein-coupled receptors termed orexin receptors -1 and -2. Orexin-A and -B are expressed by a specific population of neurons in the LHA. These neurons project to numerous brain regions, with monoaminergic and cholinergic nuclei of the hypothalamus, midbrain, and pons receiving particularly strong innervations. The orexinergic system is anatomically well-placed to coordinate the metabolic, motivational, motor, autonomic, and arousal processes necessary to elicit environmentally appropriate behaviors. Recent studies on orexin suggest that the orexinergic system plays a significant role in feeding and sleep-wakefulness regulation, possibly by coordinating the complex behavioral and physiological responses of these complementary homeostatic functions. Orexin neurons may provide an integrative link between peripheral metabolism and central regulation of behaviors required for an adaptive response to homeostatic challenges.
Sakurai T. Roles of orexins in regulation of feeding and wakefulness. Neuroreport. 2002;13(8):987-95
Orexins (hypocretins) are a novel pair of neuropeptides implicated in the regulation of energy balances and arousal. Previous reports have indicated that orexins are produced only in the lateral hypothalamic area, although orexin-containing nerve fibers were observed throughout the neuroaxis. Recent evidence shows that orexins and functional orexin receptors are found in the periphery. Vagal and spinal primary afferent neurons, enteric neurons, and endocrine cells in both the gut and pancreas display orexin- and orexin receptor-like immunoreactivity. Orexins excite secretomotor neurons in the guinea pig gut and modulate gastric and intestinal motility and secretion. In addition, orexins modulate hormone release from pancreatic endocrine cells. Moreover, fasting up-regulates the phosphorylated form of cAMP response element binding protein in orexin-immunoreactive enteric neurons, indicating a functional response to food status in these cells. The purpose of this article is to summarize evidence for the existence of a brain-gut network of orexin-containing cells that appears to play a role in the acute regulation of energy homeostasis.
Kirchgessner AL. Orexins in the brain-gut axis. Endocr Rev. 2002;23(1):1-15
Recent studies using molecular genetics in mice and dogs, as well as histopathological analyses of human disease, have come to the same conclusion: the human sleep disorder narcolepsy is caused by failure of signaling mediated by orexin (hypocretin) neuropeptides. These and other findings strongly suggest that the orexin system plays a critical role in sleep/wake regulation. In addition, the orexin system may link energy homeostasis to the regulation of sleep/wake cycles.
Mieda M, Yanagisawa M. Sleep, feeding, and neuropeptides: roles of orexins and orexin receptors. Curr Opin Neurobiol. 2002;12(3):339-45
Orexin-A-like immunoreactivity was detected in every region of human brain, but not in the pituitary. The highest concentration of orexin-A-like immunoreactivity in the human brain was found in hypothalamus (17.8 +/- 4.3 pmol/g wet weight, mean +/- SEM, n = 7), followed by thalamus, medulla oblongata, and pons. Orexin-A-like immunoreactivity was detected in the tumor tissues of ganglioneuroblastoma and neuroblastoma, but not in the tumor tissues of pheochromocytoma. Reverse phase high performance liquid chromatographic analyses of the orexin-A-like immunoreactivity in the human brain extracts and neuroblastoma extracts showed a single immunoreactive peak, which was eluted in an identical position to synthetic human orexin-A.
Arihara Z, Takahashi K, Murakami O, et al. Orexin-A in the human brain and tumor tissues of ganglioneuroblastoma and neuroblastoma. Peptides. 2000;21(4):565-70
Alterations in the hypocretin receptor 2 and preprohypocretin genes produce narcolepsy in animal models. Hypocretin was undetectable in seven out of nine people with narcolepsy, indicating abnormal hypocretin transmission.
Allen RP. Article reviewed: Hypocretin (orexin) deficiency in human narcolepsy. Sleep Med. 2000;1(2):147-148
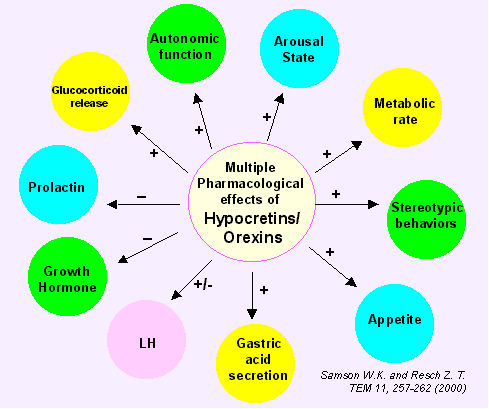
Orexin-A and B were identified as endogenous ligands for the orexin-1 (HCRT1) and orexin-2 (HCRT2) G-protein coupled receptors (Sakurai et al, 1998). These peptides are identical to two hypothalamic peptides that share a high degree of homology with secretin, designated hypocretin-1 and hypocretin-2 (De Lecea et al, 1998). ICV administration of orexin-A and B stimulated food intake in rats. Additionally, prepro-orexin mRNA levels were upregulated in fasting rats. Recent reports suggest a role in the sleep-wake cycle.
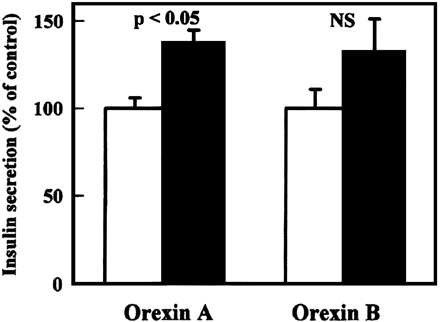
No References
| Catalog# | Product | Size | Price | Buy Now |
|---|
Social Network Confirmation