Catalog # |
Size |
Price |
|
|---|---|---|---|
| 067-29 | 10 µg | $136 |
 )
)
|
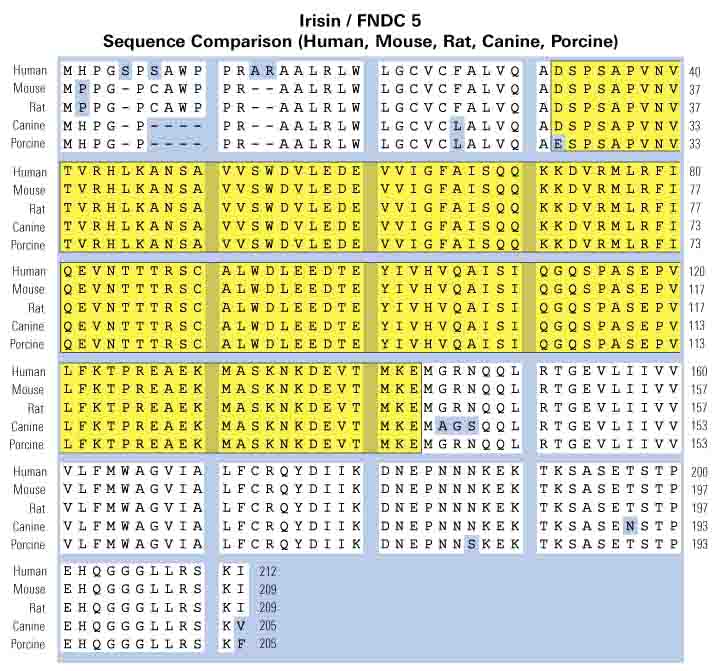
Asp-Ser-Pro-Ser-Ala-Pro-Val-Asn-Val-Thr-Val-Arg-His-Leu-Lys-Ala-Asn-Ser-Ala-Val-Val-Ser-Trp-Asp-Val-Leu-Glu-Asp-Glu-Val-Val-Ile-Gly-Phe-Ala-Ile-Ser-Gln-Gln-Lys-Lys-Asp-Val-Arg-Met-Leu-Arg-Phe-Ile-Gln-Glu-Val-Asn-Thr-Thr-Thr-Arg-Ser-Cys-Ala-Leu-Trp-Asp-Leu-Glu-Glu-Asp-Thr-Glu-Tyr-Ile-Val-His-Val-Gln-Ala-Ile-Ser-Ile-Gln-Gly-Gln-Ser-Pro-Ala-Ser-Glu-Pro-Val-Leu-Phe-Lys-Thr-Pro-Arg-Glu-Ala-Glu-Lys-Met-Ala-Ser-Lys-Asn-Lys-Asp-Glu-Val-Thr-Met-Lys-Glu
|
| 12.5 kDa | |
|
| 9037-90-5 |
|
| Exhibits correct molecular weight |
|
| Soluble in water (0.1-1.0 mg/ml). Then dilute with water or any desired aqueous buffer. |
|
|
Store in dried form at -80°C for up to 3 months. For best results, rehydrate just before use. After rehydration, keep at +4°C for 1 week or at -80°C for longer-term storage. Aliquot before freezing to avoid repeated freeze-thaw cycles. |
|
| White powder |
|
| Each vial contains 10 µg of protein. |

Abstract: Physical activity provides clinical benefit in Parkinson's disease (PD). Irisin is an exercise-induced polypeptide secreted by skeletal muscle that crosses the blood-brain barrier and mediates certain effects of exercise. Here, we show that irisin prevents pathologic alpha-synuclein (alpha-syn)-induced neurodegeneration in the alpha-syn preformed fibril (PFF) mouse model of sporadic PD. Intravenous delivery of irisin via viral vectors following the stereotaxic intrastriatal injection of alpha-syn PFF cause a reduction in the formation of pathologic alpha-syn and prevented the loss of dopamine neurons and lowering of striatal dopamine. Irisin also substantially reduced the alpha-syn PFF-induced motor deficits as assessed behaviorally by the pole and grip strength test. Recombinant sustained irisin treatment of primary cortical neurons attenuated alpha-syn PFF toxicity by reducing the formation of phosphorylated serine 129 of alpha-syn and neuronal cell death. Tandem mass spectrometry and biochemical analysis revealed that irisin reduced pathologic alpha-syn by enhancing endolysosomal degradation of pathologic alpha-syn. Our findings highlight the potential for therapeutic disease modification of irisin in PD.
Kam TI, Park H, Chou SC, et al. Amelioration of pathologic alpha-synuclein-induced Parkinson’s disease by irisin. Proc Natl Acad Sci USA. 2022;119(36):e2204835119.
Introduction/aims: Becker muscular dystrophy (BMD) is an X-linked disease leading to muscle wasting and weakness. The decrease in lean body mass (LBM) in Duchenne muscular dystrophy, has shown correlation with loss of muscle function and bone density (BD). Myokines (including irisin) are hormones secreted by skeletal muscle that allow crosstalk between muscle and bone. The present study analyzed body composition and circulating myokine levels in a cohort of BMD patients; moreover, the association between dual energy X-ray absorptiometry (DXA) parameters, functional motor assessments, and myokine levels was investigated.
Methods: All patients underwent DXA, blood samples for myokine assays, and functional motor assessments. A group of healthy controls (HCs) was also included.The authors used Phoenix's Irisin ELISA Kit (Cat. #EK-067-29) to obtain the plasma level of Irisin/FNDC5 in their research.
Barp A, Carraro E, Goggi G, et al. Body composition and myokines in a cohort of patients with Becker muscular dystrophy. Muscle and Nerve. 2022;66(1):63-70.
Background: Chronic renal failure induces bone mineral disorders and sarcopenia. Skeletal muscle affects other tissues, including bone, by releasing myokines. However, the effects of chronic renal failure on the interactions between muscle and bone remain unclear. Methods: We investigated the effects of renal failure on bone, muscle, and myokines linking muscle to bone using a mouse 5/6 nephrectomy (Nx) model. Muscle mass and bone mineral density (BMD) were analysed by quantitative computed tomography 8 weeks after Nx.
The authors used Phoenix' recombinant Irisin/FNDC5 protein (Cat. #067-29) in their research to investigate bone loss in mice.
Kawao N, Kawaguchi M, Ohira T, et al. Renal failure suppresses muscle irisin expression, and irisin blunts cortical bone loss in mice. J cachexia sarcopenia muscle. 2022;13(1):758-771.
Abstract: Limited data exist regarding the impact of an acute bout of exercise with varying intensities on irisin levels in the youth of different obesity statuses. The objectives were to (1) compare an acute bout of moderate continuous intensity (MCI) exercise and an acute bout of high-intensity interval training (HIIT) on irisin response in youth with different obesity statuses and, (2) investigate whether changes in irisin levels are correlated with exploratory outcomes. A randomized crossover design study was conducted on 25 youth aged 12–18 years old.The authors used Phoenix's Irisin ELISA kit (Cat. # EK-067-29) for finding the plasma level of Irisin/FNDC5 in their research.
Colpitts BH, Rioux BV, Eadie AL, Brunt KR, Sénéchal M. Irisin response to acute moderate intensity exercise and high intensity interval training in youth of different obesity statuses: A randomized crossover trial. Physiological Reports. 2022;10(4).
Irisin is a product of fibronectin type III domain-containing protein (Fndc5) and is involved in the regulation of adipokine secretion and the differentiation of osteoblasts and osteoclasts. In this study, we aimed to determine whether irisin lacking affects glucose/lipid and bone metabolism. We knocked out the Fndc5 gene to generate irisin-lacking mice. Remarkable, irisin lacking was related to poor ‘browning response’, with a bigger size of the intraperitoneal white adipose cell and decreased a number of brown adipose cells in brown adipose of interscapular tissue. The irisin lacking mice had hyperlipidemia and insulin resistance, reduced HDL-cholesterol level, increased LDL-cholesterol level, and decreased insulin sensitivity. The lacking of irisin was associated with reduced bone strength and bone mass in mice. The increased number of osteoclasts and higher expression of RANKL indicated increased bone resorption in irisin lacking mice. The level of IL-6 and TNF-α also increased in irisin lacking mice. The results showed that irisin lacking was related to decreased ‘browning response’, glucose/lipid metabolic derangement, and reduced bone mass with increased bone resorption. Further studies are needed to confirm these initial observations and explore the mechanisms underlying the effects of irisin on glucose/lipid and bone metabolism.Luo Y, Qiao X, Ma Y, Deng H, Xu CC, Xu L. Disordered metabolism in mice lacking irisin. Sci Rep. 2020;10(1):17368.
Irisin, a recently discovered myokine, has been considered a prognostic factor in several cardiovascular diseases. Nevertheless, no data are available on the role of irisin in patients with heart failure (HF), both with preserved (HFpEF) or reduced (HFrEF) ejection fraction. We have therefore evaluated the circulating irisin levels in HFpEF and HFrEF patients, correlating them with metabolic parameters and total antioxidant capacity (TAC), as index of oxidative stress. Irisin was significantly higher in HFpEF than in HFrEF patients (7.72 ± 0.76 vs 2.77 ± 0.77 ng/ml, respectively). An inverse correlation between irisin and TAC was found in HFpEF, but not in HFrEF. Conversely, no correlation was present with HOMA index. These data support the hypothesis that a different pathophysiological mechanism is involved in the two HF subtypes, and oxidative stress modulates irisin secretion.This publication used the irisin ELISA kit (EK-067-29) from Phoenix Pharmaceuticals to measure the plasma level of irisin.
Silvestrini A, Bruno C, Vergani E, et al. Circulating irisin levels in heart failure with preserved or reduced ejection fraction: A pilot study. PLoS ONE. 2019;14(1):e0210320.
Defective brain hormonal signaling has been associated with Alzheimer's disease (AD), a disorder characterized by synapse and memory failure. Irisin is an exercise-induced myokine released on cleavage of the membrane-bound precursor protein fibronectin type III domain-containing protein 5 (FNDC5), also expressed in the hippocampus. Here we show that FNDC5/irisin levels are reduced in AD hippocampi and cerebrospinal fluid, and in experimental AD models. Knockdown of brain FNDC5/irisin impairs long-term potentiation and novel object recognition memory in mice. Conversely, boosting brain levels of FNDC5/irisin rescues synaptic plasticity and memory in AD mouse models. Peripheral overexpression of FNDC5/irisin rescues memory impairment, whereas blockade of either peripheral or brain FNDC5/irisin attenuates the neuroprotective actions of physical exercise on synaptic plasticity and memory in AD mice. By showing that FNDC5/irisin is an important mediator of the beneficial effects of exercise in AD models, our findings place FNDC5/irisin as a novel agent capable of opposing synapse failure and memory impairment in AD.Lourenco MV, Frozza RL, De freitas GB, et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer's models. Nat Med. 2019;25(1):165-175.
Half a decade ago, transmembrane protein fibronectin type III domain-containing protein 5 (FNDC5) was found to be cleaved as a novel myokine irisin, which burst into prominence for browning of white adipose tissue during exercise. However, FNDC5, the precursor of irisin, has been paid relatively little attention compared with irisin despite evidence that FNDC5 is associated with the metabolic syndrome, which accounts for one-fourth of the world's adult population and contributes to diabetes, cardiovascular disease and all-cause mortality. Besides N-terminal and C-terminal sequences, the FNDC5 protein contains an irisin domain and a short transmembrane region. FNDC5 has shown to be widely distribute in different tissues and is highly expressed in heart, brain, liver, and skeletal muscle. Clinical studies have demonstrated that FNDC5 is essential for maintaining metabolic homeostasis and dysregulation of FNDC5 will lead to systemic metabolism imbalance and the onset of metabolic disorders. Growing evidence has suggested that FNDC5 gene polymorphisms are related to health and disease in different human populations. Additionally, FNDC5 has been found relevant to the regulation of metabolism and metabolic syndrome through diverse upstream and downstream signaling pathways in experimental studies. The present review summarizes the characteristics, clinical significance, and molecular mechanisms of FNDC5 in metabolic syndrome and proposes a novel concept that FNDC5 is activated by forming a putative ligand-receptor complex. Knowledge about the role of FNDC5 may be translated into drug development and clinical applications for the treatment of metabolic disorders.Cao RY, Zheng H, Redfearn D, Yang J. FNDC5: A novel player in metabolism and metabolic syndrome. Biochimie. 2019;158:111-116.
Irisin is secreted by muscle, increases with exercise, and mediates certain favorable effects of physical activity. In particular, irisin has been shown to have beneficial effects in adipose tissues, brain, and bone. However, the skeletal response to exercise is less clear, and the receptor for irisin has not been identified. Here we show that irisin binds to proteins of the αV class of integrins, and biophysical studies identify interacting surfaces between irisin and αV/β5 integrin. Chemical inhibition of the αV integrins blocks signaling and function by irisin in osteocytes and fat cells. Irisin increases both osteocytic survival and production of sclerostin, a local modulator of bone remodeling. Genetic ablation of FNDC5 (or irisin) completely blocks osteocytic osteolysis induced by ovariectomy, preventing bone loss and supporting an important role of irisin in skeletal remodeling. Identification of the irisin receptor should greatly facilitate our understanding of irisin's function in exercise and human health.Kim H, Wrann CD, Jedrychowski M, et al. Irisin Mediates Effects on Bone and Fat via αV Integrin Receptors. Cell. 2018;175(7):1756-1768.e17.
Objective: The new adipokine, which is asprosin, affects glucose release from the liver to the blood, and thus, influences exercise metabolism. This is the first study assessing whether single anaerobic exercise affects asprosin secretion in women and men.Methods: 10 men and 10 women (aged 21.64 ± 1.22 and 22.64 ± 1.49, respectively) performed a single 20-s bicycle sprint. Blood samples were collected before exercise and in the 3', 15', 30', and 60' of recovery, and 24 h after competition.Results: Only in women did asprosin (P = 0.001) (15', 30', 60', and 24 h after exercise) and irisin (P < 0.001) (15', 30', and 60') concentrations increase. Leptin, however, decreased (P = 0.001) at 3', 15', and 30' in women. There was an increase in interleukin-6 (P < 0.001) at 3', 15', 30', and 60' of recovery in men, at 15', 30', 60', and 24 h of recovery in women, along with a simultaneous decrease in interleukin-1β (P < 0.001) at 15', 30', and 60' of recovery in men, and at 15' and 30' of recovery in women (r = -0.35, P < 0.001). There was a positive correlation between asprosin and adiponectin and a negative one between asprosin and leptin. The increase in irisin concentration at 30' of recovery was positively correlated with the increase in asprosin concentration and percentage fat content, while being negatively correlated with total and lean body mass (LBM).Conclusion: The single anaerobic effort induced an increase in asprosin and irisin secretion while reducing leptin secretion in women. Adipocytokine concentration changes are inter-related. Regardless of sex, anaerobic efforts induce anti-inflammatory effects.This publication used the irisin EIA kit (EK-067-16) from Phoenix Pharmaceuticals to measure the serum level of irisin.
He L, He WY, A LT, Yang WL, Zhang AH. Lower Serum Irisin Levels Are Associated with Increased Vascular Calcification in Hemodialysis Patients. Kidney Blood Press Res. 2018;43(1):287-295.
Epicardial fat located adjacent to the heart and coronary arteries is associated with increased cardiovascular risk. Irisin is a myokine produced by skeletal muscle after physical exercise, and originally described as a molecule able to promote the browning of white adipose tissue and energy expenditure. In order to decrease cardiovascular risk, it has been proposed as a promising therapeutic target in obesity and type 2 diabetes. We investigated the relationships between serum concentrations of irisin and the adipokines adiponectin and leptin and body fat including epicardial fat in patients undergoing cardiovascular surgery. We obtained serum samples from 93 patients undergoing cardiovascular surgery (age 69.6 (SD 12.8) years, BMI 24.1 ± 4.8 kg/m2). Computed tomography (CT) and echocardiographic data were obtained from the routine preoperative examination. Subcutaneous fat area (SFA, cm2) and visceral fat area (VFA, cm2) near the umbilicus were automatically measured using the standard fat attenuation range. Epicardial fat area (EFA, cm2) was measured at the position where the heart became a long axis image with respect to the apex of the heart in the coronal section image. Total body fat mass, body fat percentage, and skeletal muscle volume (SMV) were estimated using bioelectrical impedance analysis (BIA). Serum irisin concentration was measured by enzyme-linked immunosorbent assay, and compared with adiponectin and leptin concentrations. The data were also compared with the clinical biochemical data. EFA was strongly correlated with BMI (P = 0.0001), non-HDL-C (P = 0.029), TG (P = 0.004), body fat mass (P = 0.0001), and body fat percentage (P = 0.0001). Serum leptin concentration showed a significant positive correlation with BMI (P = 0.0001) and TG (P = 0.001). Adiponectin, but not irisin, showed a significant negative correlation with BMI (P = 0.006) and TG (P = 0.001). Serum leptin level had a significant positive correlation with EFA, VFA, and SFA. In contrast, the serum adiponectin level was significantly negatively correlated with EFA, VFA, and SFA. The serum irisin level was also negatively correlated with EFA (r = -0.249, P = 0.015), and SFA (r = -0.223, P = 0.039), and tended to correlate with VFA (r = -0.198, P = 0.067). The serum level of adiponectin was negatively correlated with that of leptin (r = -0.296, P = 0.012), but there were no significant correlations between irisin and either adiponectin or leptin. Multivariate linear regression demonstrated that EFA showed a positive association with serum leptin level (β = 0.438, P = 0.0001) and a negative correlation with serum irisin level (β = -0.204, P = 0.038) and serum adiponectin level (β = -0.260, P = 0.015) after adjusting for age, sex, and BMI. The present study provided the first evidence of associations of the serum irisin and adipokines (adiponectin and leptin) concentrations with epicardial fat in cardiovascular surgery patients. Irisin may play a role in preventing excess adiposity including epicardial fat, and consequently cardiovascular risk in patients.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for irisin serum level assay.
Kaneda H, Nakajima T, Haruyama A, et al. Association of serum concentrations of irisin and the adipokines adiponectin and leptin with epicardial fat in cardiovascular surgery patients. PLoS ONE. 2018;13(8):e0201499.
Irisin, an adipomyokine identified in 2012, has been investigated in association with common pregnancy complications, including gestational diabetes mellitus, preeclampsia, and intrauterine growth restriction. The objective of this study is to examine the potential role of irisin in preterm birth (PTB) by comparing its level between mothers with term and preterm labor. Maternal peripheral blood and cord blood samples were collected from 30 mothers who delivered prematurely and from 35 mothers who delivered at term. Irisin concentrations were measured in all samples using ELISA, and four common single nucleotide polymorphisms in the irisin gene were determined (rs16835198, rs726344, rs3480, and rs1746661). Univariable and multivariable regression modeling was applied to evaluate maternal and cord blood irisinconcentrations in relation to preterm/term labor. Irisin concentration in umbilical cord blood was found to be associated with PTB in the univariable model (p = 0.046). On the other hand, no differences in maternal blood irisin levels between mothers with preterm and term deliveries were established. To the best of our knowledge, this is the first study determining irisin levels in term and preterm deliveries in maternal peripheral blood and umbilical cord blood. Our study shows a possible association between cord blood irisin concentration and PTB occurrence.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for irisin plasma level assay.
Pavlova T, Zlamal F, Tomandl J, Hodicka Z, Gulati S, Bienertova-vasku J. Irisin Maternal Plasma and Cord Blood Levels in Mothers with Spontaneous Preterm and Term Delivery. Dis Markers. 2018;2018:7628957.
BACKGROUND: Irisin is a recently identified exercise-induced hormone that stimulates the "browning" of the white adipose tissue, at least in mice. In chronic kidney disease (CKD) patients, irisin regulation is not fully understood, and little attention has been given to the effects of exercise on irisin levels in these patients. The purpose of this study was to assess the effects of high intensity exercise on irisin plasma levels in CKD patients under hemodialysis (HD).METHODS: Fifteen HD patients (5 men, 44.4 ± 15.1 years old) were studied and served as their own controls. High intensity (single session) intradialytic strength exercises consisted of three sets of ten repetitions with four different movements in both lower limbs during 30 minutes. Blood samples were collected on different days (exercise and non-exercise day) at exactly the same time (30 and 60 minutes after the start of dialysis session). Plasma irisin levels were measured by ELISA assay and anthropometric and biochemical parameters were evaluated.RESULTS: Irisin plasma levels were significantly reduced in both exercise day (125.0 ± 18.5 to 117.4 ± 15.0 ng/mL, p=0.02) and non-exercise day (121.5 ± 13.7 to 115.4 ± 17.2 ng/mL, p=0.02) after 60 minutes of dialysis.CONCLUSION: These data suggest that intense intradialytic strength exercise was unable to increase the circulating concentration of irisin in HD patients. Moreover, our data show that after one hour of dialysis session, irisin plasma levels may be reduced.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for irisin serum level assay.
Esgalhado MGBM, Stockler-pinto MB, Cardozo LFMF, Barboza JE, Mafra D. Does high intensity exercise affects irisin plasma levels in hemodialysis patients? A pilot study. J Bras Nefrol. 2018;40(1):53-58.
BACKGROUND/AIMS: All-trans retinoic acid (ATRA) has protective effects against obesity and metabolic syndrome. We here aimed to gain further insight into the interaction of ATRA with skeletal muscle metabolism and secretory activity as important players in metabolic health.METHODS: Cultured murine C2C12 myocytes were used to study direct effects of ATRA on cellular fatty acid oxidation (FAO) rate (using radioactively-labelled palmitate), glucose uptake (using radioactively-labelled 2-deoxy-D-glucose), triacylglycerol levels (by an enzymatic method), and the expression of genes related to FAO and glucose utilization (by RT-real time PCR). We also studied selected myokine production (using ELISA and immunohistochemistry) in ATRA-treated myocytes and intact mice.RESULTS: Exposure of C2C12 myocytes to ATRA led to increased fatty acid consumption and decreased cellular triacylglycerol levels without affecting glucose uptake, and induced the expression of the myokine irisin at the mRNA and secreted protein level in a dose-response manner. ATRA stimulatory effects on FAO-related genes and the Fndc5 gene (encoding irisin) were reproduced by agonists of peroxisome proliferator-activated receptor β/δ and retinoid X receptors, but not of retinoic acid receptors, and were partially blocked by an AMP-dependent protein kinase inhibitor. Circulating irisin levels were increased by 5-fold in ATRA-treated mice, linked to increased Fndc5 transcription in liver and adipose tissues, rather than skeletal muscle. Immunohistochemistry analysis of FNDC5 suggested that ATRA treatment enhances the release of FNDC5/irisin from skeletal muscle and the liver and its accumulation in interscapular brown and inguinal white adipose depots.CONCLUSION: These results provide new mechanistic insights on how ATRA globally stimulates FAO and enhances irisin secretion, thereby contributing to leaning effects and improved metabolic status.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for circulating irisin level measurement.
Amengual J, García-carrizo FJ, Arreguín A, et al. Retinoic Acid Increases Fatty Acid Oxidation and Irisin Expression in Skeletal Muscle Cells and Impacts Irisin In Vivo. Cell Physiol Biochem. 2018;46(1):187-202.
BACKGROUND/AIMS: Vascular calcification, which involves an active cellular transformation of vascular smooth muscle cells into bone forming cells, is prevalent and predicts mortality in dialysis patients. Its mechanisms are complex and unclear. We presume that irisin, a newly identified myokine also may play roles in vascular calcification in hemodialysis patients. This study aims to evaluate serum irisin levelsand establish their relation to vascular calcification and other parameters in hemodialysis patients.METHODS: A total of 150 patients on maintenance hemodialysis treatment and 38 age- and sex-matched healthy controls were enrolled in this cross-sectional study. Serum irisin concentrations were measured by ELISA. Vascular calcification was evaluated by abdominal aortic calcification scores.RESULTS: Serum irisin concentrations were significantly lower in hemodialysis patients than in controls [52.8 (22.0, 100.0) vs. 460.8 (434.8, 483.4) ng/ml, P<0.01]. In addition, irisin was negatively correlated with the parathyroid hormone level (P=0.01). The HD patients with vascular calcification showed significantly lower serum irisin concentrations [39.0 (21.7, 86.2) vs.79.0 (39.5, 130.2) ng/mL, P<0.01]. Compared with the group without vascular calcification multivariate logistic regression analyses revealed that serum irisin, HD vintage and age were significant independent determinant factors for vascular calcification in HD patients.CONCLUSION: Our results are the first to provide a clinical evidence of the association between serum irisin and vascular calcification in HD patients. Lower irisin levels, long-term hemodialysis and old ages are independent risk factors in HD patients.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for irisin serum level assay.
He L, He WY, A LT, Yang WL, Zhang AH. Lower Serum Irisin Levels Are Associated with Increased Vascular Calcification in Hemodialysis Patients. Kidney Blood Press Res. 2018;43(1):287-295.
The mechanisms underlying the metabolic improvements following aerobic exercise training remain poorly understood. The primary aim of this study was to determine if an adipomyokine, irisin, responded to acute exercise was associated with the metabolic adaptations to chronic aerobic exercise in obese youth. The acute response to exercise was assessed in 11 obese youth following 45-min acute bouts of aerobic (AE) and resistance exercise (RE). The irisin area under the curve (pre-exercise, 15, 30, and 45 min) during these AE sessions were the main exposure variables. The primary outcome measure was the change in insulin sensitivity using the Matsuda index, following 6 weeks of RE training, delivered for 45 min, three times per week at 60-65% 1RM. Participants were also categorized as either responders (above) or nonresponders (below) based on the percentage change in the Matsuda index following the 6-week intervention. Irisin increased significantly during the acute bout of AE from 29.23 ± 6.96 to 39.30 ± 7.05 ng/mL; P = 0.028, but not significantly during the RE session (P = 0.182). Absolute and relative change in irisin during the acute bout of AE was associated with absolute and relative change in Matsuda index (r = 0.68; P = 0.022 and r = 0.63; P = 0.037) following the 6-week RE intervention. No such association was observed with the irisin responseto acute RE (all P > 0.05). Responders to the 6-week RE intervention displayed a fourfold greater irisin response to acute AE (90.0 ± 28.0% vs. 22.8 ± 18.7%; P = 0.024) compared to nonresponders. Irisin increases significantly following an acute bout of AE, but not RE, and this response is associated with a greater improvement in insulin sensitivity in response to chronic resistance training.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for quantification of human irisin plasma level.
Devin R. Blizzard LeBlanc, Brittany V. Rioux, Cody Pelech et al., Physiol Rep. 2017 Dec; 5(23): e13539.
Irisin is a myokine encoded in its precursor fibronectin type III domain containing 5 (FNDC5). It is abundantly expressed in cardiac and skeletal muscle, and is secreted upon the activation of peroxisome proliferator-activated receptor gamma coactivator-1 (PGC-1 alpha). We aimed to study the role of irisin on cardiac function and muscle protein regulation in zebrafish. Western blot analyses detected the presence of irisin protein (23 kDa) in zebrafish heart and skeletal muscle, and irisin immunoreactivity was detected in both tissues. Irisin siRNA treated samples did not show bands corresponding to irisin in zebrafish. In vitro studies found that treatment with irisin (0.1 nM) downregulated the expression of PGC-1 alpha, myostatin a, and b, while upregulating troponin C mRNA expression in zebrafish heart and skeletal muscle. Exogenous irisin (0.1 and 1 ng/g B.W) increased diastolic volume, heart rate and cardiac output, while knockdown of irisin (10 ng/g B.W) showed opposing effects on cardiovascular function. Irisin (1 and 10 ng/g B.W) downregulated PGC-1 alpha, myostatin a and b, and upregulated troponin C and troponin T2D mRNA expression. Meanwhile, knockdown of irisin showed opposing effects on troponin C, troponin T2D and myostatin a and b mRNAs in zebrafish heart and skeletal muscle. Collectively, these results identified muscle proteins as novel targets of irisin, and added irisin to the list of peptide modulators of cardiovascular physiology in zebrafish.
This publication used the irisin antibody (H-067-17) and c-terminal peptide (067-17) from Phoenix Pharmaceuticals for in vivo and in vitro assays.
Sundarrajan L, Yeung C, Hahn L, Weber LP, Unniappan S. Irisin regulates cardiac physiology in zebrafish. PLoS ONE. 2017;12(8):e0181461.
Type 2 diabetes mellitus (T2DM) is caused by insulin resistance and β cell dysfunction. In recent studies reported that several markers associated with insulin sensitivity in skeletal muscle, Adiponectin and other parameters, such as fatty acid-binding protein (FABP4), have been reported to regulate insulin resistance, but it remains unclear which factor mostly affects insulin resistance in T2DM. In this cross-sectional study, we evaluated the relationships between several kinds of biomarkers and insulin resistance, and insulin secretion in T2DM and healthy controls. We recruited 30 participants (12 T2DM and 18 non-diabetic healthy controls). Participants underwent a meal tolerance test during which plasma glucose, insulin and serum C-peptide immunoreactivity were measured. We performed a hyperinsulinemic-euglycemic clamp and measured the glucose-disposal rate (GDR). The fasting serum levels of adiponectin, insulin-like growth factor-1, irisin, autotaxin, FABP4 and interleukin-6 were measured by ELISA. We found a strong negative correlation between FABP4 concentration and GDR in T2DM (r = -0.657, p = 0.020). FABP4 also was positively correlated with insulin secretion during the meal tolerance test in T2DM (IRI (120): r = 0.604, p = 0.038) and was positively related to the insulinogenic index in non-DM subjects (r = 0.536, p = 0.022). Autotaxin was also related to GDR. However, there was no relationship with insulin secretion. We found that serum FABP4 concentration were associated with insulin resistance and secretion in T2DM. This suggests that FABP4 may play an important role in glucose homeostasis.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals.
Nakamura R, Okura T, Fujioka Y, et al. Serum fatty acid-binding protein 4 (FABP4) concentration is associated with insulin resistance in peripheral tissues, A clinical study. PLoS ONE. 2017;12(6):e0179737.
BACKGROUND: Irisin is a recently discovered myokine thought to be involved in multiple metabolism abnormalities in most dialysis patients. However, the myokine has not been thoroughly studied in peritoneal dialysis. This study aimed to evaluate serum irisin levels and establish their relation to dialysis adequacy, insulin resistance, and bone metabolism status in patients on peritoneal dialysis.METHODS: A total of 59 nondiabetic prevalent peritoneal dialysis patients and 52 age- and sex-matched healthy controls were enrolled in this cross-sectional study. Serum irisin concentration was assessed by enzyme-linked immunosorbent assay. The correlations between serum irisin and dialysis adequacy, clinical, and metabolic variables were investigated.RESULTS: Serum irisin levels were lower in nondiabetic peritoneal dialysis patients (17.02ng/ml) compared with healthy controls (22.17ng/ml, P<0.001). Multivariate regression analysis revealed that fasting glucose levels were correlated inversely with serum irisin levels in peritoneal dialysis patients. Serum irisin levels were associated with neither insulin resistance nor bone metabolism in our patients. Serum irisin levels were positively associated with peritoneal Kt/Vurea (β = 4.933, 95% confidence interval [CI] = 0.536-9.331, P = 0.029) and peritoneal CCr (β = 0.259, 95% CI = 0.053-0.465, P = 0.015) among peritoneal dialysis patients.CONCLUSIONS: The study demonstrated that non-diabetic peritoneal dialysis patients have lower serum irisin levels, and the levels were correlated with peritoneal dialysis adequacy, indicating adequate dialysis may improve irisin secretion. Additional studies are needed to provide a confirmation.
This publication used the irisin EIA kit (EK-067-29) from Phoenix Pharmaceuticals for irisin serum level assay.
Tan Z, Ye Z, Zhang J, et al. Serum irisin levels correlated to peritoneal dialysis adequacy in nondiabetic peritoneal dialysis patients. PLoS ONE. 2017;12(4):e0176137.
Exercise provides many health benefits, including improved metabolism, cardiovascular health, and cognition. We have shown previously that FNDC5, a type I transmembrane protein, and its circulating form, irisin, convey some of these benefits in mice. However, recent reports questioned the existence of circulating human irisin both because human FNDC5 has a non-canonical ATA translation start and because of claims that many human irisin antibodies used in commercial ELISA kits lack required specificity. In this paper we have identified and quantitated human irisin in plasma using mass spectrometry with control peptides enriched with heavy stable isotopes as internal standards. This precise state-of-the-art method shows that human irisin is mainly translated from its non-canonical start codon and circulates at ∼ 3.6 ng/ml in sedentary individuals; this level is increased to ∼ 4.3 ng/ml in individuals undergoing aerobic interval training. These data unequivocally demonstrate that human irisin exists, circulates, and is regulated by exercise.
Jedrychowski MP, Wrann CD, Paulo JA, et al. Cell Metab. 2015;22(4):734-740.
As a link between exercise and metabolism, irisin is assumed to be involved in increased total body energy expenditure, reduced body weight, and increased insulin sensitivity. Although our recent evidence supported the contribution of irisin to vascular endothelial cell (ECs) proliferation and apoptosis, further research of irisin involvement in the angiogenesis of ECs was not conclusive. In the current study, it was found that irisinpromoted Human Umbilical Vein Endothelial Cell (HUVEC) angiogenesis via increasing migration and tube formation, and attenuated chemically-induced intersegmental vessel (ISV) angiogenic impairment in transgenic TG (fli1: GFP) zebrafish. It was further demonstrated that expression of matrix metalloproteinase (MMP) 2 and 9 were also up-regulated in endothelial cells. We also found that irisin activated extracellular signal-related kinase (ERK) signaling pathways. Inhibition of ERK signaling by using U0126 decreased the pro-migration and pro-angiogenic effect of irisin on HUVEC. Also, U0126 inhibited the elevated expression of MMP-2 and MMP-9 when they were treated with irisin. In summary, these findings provided direct evidence that irisin may play a pivotal role in maintaining endothelium homeostasis by promoting endothelial cell angiogenesis via the ERK signaling pathway.
Wu F, Song H, Zhang Y et al., PLoS One. 2015 Aug 4;10(8):e0134662. doi: 10.1371/journal.pone.0134662. eCollection 2015
Objective: Betatrophin has recently been introduced as a novel adipokine/hepatokine, which promotes pancreatic β cell proliferation and improves glucose tolerance in several mouse models of insulin resistance. However, regulation of betatrophin in gestational diabetes mellitus (GDM), as well as its association with markers of obesity, such as glucose and lipid metabolism, inflammation, and renal function, have not been elucidated.
Design and methods: Circulating betatrophin was quantified in 74 women with GDM and 74 healthy and gestational age-matched controls by ELISA. In a subset of the study population comprising of 85 patients (41 previous controls, 44 previous women with GDM), postpartum betatrophin levels were measured in a follow-up study.
Results: Median (interquartile range) serum betatrophin levels were higher in women with GDM (1.79 (0.53) ?g/l) as compared to non-diabetic pregnant controls (1.58 (0.44) ?g/l) (P=0.002). In multivariate analysis, GDM status was an independent and positive predictor of circulating betatrophin (P=0.001). Furthermore, betatrophin levels were significantly higher during gestation (1.70 (0.53) ?g/l) as compared to postpartum levels (1.55 (0.66) ?g/l) (P=0.028). Moreover, postpartum irisin remained a positive and independent predictor of postpartum betatrophin concentrations.
Conclusions: Women with GDM have significantly higher betatrophin levels as compared to healthy pregnant controls and GDM status positively predicts circulating betatrophin. Furthermore, postpartum levels are significantly lower as compared to betatrophin concentrations during pregnancy. Moreover, irisin is a significant predictor of postpartum betatrophin levels.
This publication used an Irisin kit from Phoenix Pharmaceuticals.
Thomas Ebert, Susan Kralisch, Ulrike Wurst et al., Eur. J. Endocrinol., Jul 2015; 173: 1 - 7
Irisin is a newly identified myokine. Several studies have reported irisin concentrations in patients with gestational diabetes mellitus (GDM), but because of smaller sample sizes, the data from previous reports showed a wide range in serum/plasma irisin. Therefore, the present investigation is designed to summarize a precise confidence interval of circulating irisin in participants with GDM from a cross-sectional study in Chinese population and a meta-analysis for validation. Serum irisin was tested in patients with GDM and healthy controls (newly diagnosed cases: 61 and matched controls: 61) in the cross-sectional study. The two groups of participants were matched with > for age and pregnancy duration. Furthermore, we did a comprehensive meta-analysis to confirm whether serum/plasma irisin differs between participants with GDM and controls. Articles reported “circulating irisin and GDM” in Medline, PubMed, and EMBase were obtained, with the key word “myokine” or “irisin”. The comparison was analyzed by Review Manager 5.2. In the cross-sectional investigation, serum irisin showed a significant lower level in the GDM patients, compared with that in the control group. In the meta-analysis study, the summarized results of the present 5 studies in which 632 participants were included indicated that there was a lower level irisin of -58.68 ng/ml [95% confidence interval (CI)](-113.42, -3.93, P=0.04) in GDM patients than in the control group. The present cross-sectional investigation and meta-analysis is the first to show significant lower circulating irisin in subjects with GDM.
This publication used an Irisin kit from Phoenix Pharmaceuticals.
Zhao L, Li J, Li ZL et al., Endocr J. 2015 Jul 29. [Epub ahead of print]
Vascular complications are the major causes of death in patients with diabetes, and endothelial dysfunction is the earliest event in vascular complications of diabetes. It has been reported that plasma irisin level is significantly reduced in patients with type 2 diabetic patients. The present study aimed to investigate whether irisin improved endothelial function in type 2 diabetes as well as the underlying mechanisms. The type 2 diabetes model was established by feeding C57BL/6 mice with high-fat diet. The type 2 diabetic mice exhibited reduced serum irisin level and impaired endothelial function. Irisin treatment (0.5mg/kg/d) for two weeks improved vascular function based on the evaluation of endothelium-dependent vasorelaxation and p-VASP levels. To investigate the direct endothelial protective effects of irisin, diabetic aortic segments were incubated with irisin(1?g/ml) ex vivo. Exposure to irisin improved endothelium-dependent vasorelaxation of diabetic aortas. Mechanically, the diabetic aortic segments exhibited increased oxidative/nitrative stresses. Irisin reduced the diabetes-induced oxidative/nitrative stresses evidenced by reducing overproduction of superoxide and peroxynitrite, and down-regulation of iNOS and gp91phox. To further investigate the protective effects of irisin on endothelial cells and the underlying mechanisms, human umbilical vein endothelial cells (HUVECs) cultured in high-glucose/high-fat (HG/HF) medium were pre-incubated with irisin. Irisin (1?g/ml) reduced the oxidative/nitrative stresses and apoptosis induced by HG/HF in HUVECs probably via inhibiting activation of PKC-?/NADPH oxidase and NF-?B/iNOS pathways. Taken together, irisin alleviates endothelial dysfunction in type 2 diabetes partially via reducing oxidative/nitrative stresses through inhibiting signaling pathways implicating PKC-?/NADPH oxidase and NF-?B/iNOS, suggesting that irisin may be a promising molecule for the treatment of vascular complications of diabetes.
Zhu D, Wang H, Zhang J et al., J Mol Cell Cardiol. 2015 Jul 27. pii: S0022-2828(15)30020-1. doi: 10.1016/j.yjmcc.2015.07.015. [Epub ahead of print]
Irisin is a newly identified hormone induced in muscle and adipose tissues by physical activity. This protein and its encoding gene have been identified in the brain; in addition, the precursor for irisin, FNDC5, can cross the blood-brain barrier. The fact that irisin is secreted during exercise together with the lower resting heart rate in athletes prompted us to investigate the effect of irisin on cardiac-projecting vagal neurons of nucleus ambiguus, a key regulatory site of heart rate. In vitro experiments in cultured nucleus ambiguus neurons indicate that irisin activates these neurons, inducing an increase in cytosolic Ca(2+) concentration and neuronal depolarization. In vivo microinjection of irisin into the nucleus ambiguuspromotes bradycardia in conscious rats. Our study is the first to report the effects of irisin on the neurons controlling the cardiac vagal tone and to link a myokine to a cardioprotective role, by modulating central cardiovascular regulation.
This publication used an Irisin recombinant protein from Phoenix Pharmaceuticals.
Brailoiu E, Deliu E, Sporici RA, Brailoiu GC, Physiol Rep. 2015 Jun;3(6). pii: e12419. doi: 10.14814/phy2.12419
INTRODUCTION: Irisin is a newly identified 112 amino acid hormone, derived as a product of fibronectin type III domain containing 5 (FNDC5), which is highly related to metabolic activity in skeletal muscle and brown fat. The effects of irisin on cardiovascular functions are unknown.
PURPOSE: To explore the effects of central and peripheral irisin on cardiovascular functions.
METHODS: Irisin was either administrated into 3rd ventricle of rats or intravenously, and its effects on blood pressure and cardiac contractibility measured.
RESULTS: Administration of recombinant human irisin into the 3rd brain ventricle of rats activated neurons in the paraventricular nuclei of the hypothalamus. Central administration of irisin increased blood pressure and cardiac contractibility. Exogenous irisin reversed atenolol-induced inhibition of cardiac contractibility. In contrast, peripheral administration of irisin reduced blood pressure in both control and spontaneously hypertensive rats. Irisin dilated mesenteric artery rings through ATP-sensitive potassium channels.
CONCLUSION: Our studies indicate that central and peripheral irisin may differentially regulate cardiovascular activities.
This publication used an Irisin recombinant protein from Phoenix Pharmaceuticals.
Zhang W, Chang L, Zhang C et al., Cardiovasc Drugs Ther. 2015 Mar 31. [Epub ahead of print]
Irisin is a novel myokine produced by skeletal muscle. However, its metabolic role is poorly understood. In the present study, irisin induced glucose uptake in differentiated skeletal muscle cells. It increased AMP-activated protein kinase (AMPK) phosphorylation and the inhibition of AMPK blocked glucose uptake. It also increased reactive oxygen species (ROS) generation. N-acetyl-cysteine (NAC), a ROS scavenger, blocked irisin-induced AMPK phosphorylation. Moreover, irisin activated p38 mitogen-activated protein kinase (MAPK) in an AMPK-dependent manner. The inhibition and knockdown of p38 MAPK blocked irisin-induced glucose uptake. A colorimetric absorbance assay showed that irisin stimulated the translocation of GLUT4 to the plasma membrane, and that this effect was suppressed in cells pre-treated with a p38 MAPK inhibitor or p38 MAPK siRNA. In primary cultured myoblast cells, irisin increased the concentration of intracellular calcium. STO-609, a calcium/calmodulin-dependent protein kinase kinase (CaMKK) inhibitor, blocked irisin-induced AMPK phosphorylation, implying that calcium is involved in irisin-mediated signaling. Our results suggest that irisin plays an important role in glucose metabolism via the ROS-mediated AMPK pathway in skeletal muscle cells.
This publication used an Irisin recombinant protein from Phoenix Pharmaceuticals.
Jeong Lee H, Ok Lee J, Kim N et al., Mol Endocrinol. 2015 Mar 31:me20141353. [Epub ahead of print]
To elucidate the effects of endurance training on circulating irisin levels in young and middle-aged/older adults, and to determine the association between endurance training-induced alteration of irisin and reduction in body fat. Twenty-five healthy young (age 21 ± 1 years; 16 men, 9 women) and 28 healthy middle-aged/older adults (age 67 ± 8 years; 12 men, 16 women) participated in the study. Each age cohort was divided into two groups: the endurance-training group (14 young, 14 middle-aged/older) and the control group. Subjects in the training groups completed an 8-week endurance-training program (cycling at 60-70% peak oxygen uptake [[Formula: see text]O2peak] for 45 min, 3 days/week). Before and after the intervention, we evaluated serum irisin level, [Formula: see text]O2peak, and body composition. The increase in [Formula: see text]O2peak in the young and middle-aged/older training groups after the intervention period was significantly greater than those in the young and middle-aged/older control groups (P < 0.05). Serum irisin level was significantly increased in the middle-aged/older training group after the intervention period (P < 0.01), but not in the young training group. Furthermore, in the middle-aged/older training group, the endurance training-induced reduction in visceral adipose tissue area was negatively correlated with the change in serum irisin level (r = -0.54, P < 0.05). These results suggest a possible role for secreted irisin in the exercise-induced alteration of abdominal visceral fat in middle-aged and older adults.
Miyamoto-Mikami E, Sato K, Kurihara T et al., PLoS One. 2015 Mar 20;10(3):e0120354. doi: 10.1371/journal.pone.0120354
Irisin is a newly discovered factor that is secreted by skeletal muscle and plays an important role in the homeostasis and metabolism of energy balance. This study used irisin radiolabeled with 125I and small-animal SPECT/CT imaging to investigate the metabolic elimination and distribution ofirisin in vivo. Irisin was labeled with 125I using the Iodogen method. Small-animal SPECT/CT imaging was performed on C57/B16 mice at 15, 30, 60, 120, and 240?min after receiving a tail vein injection, and the radioactive distribution in the organs of mice was determined at 15, 60, and 120?min. Small-animal SPECT/CT imaging revealed the highest level of radioactivity in the gallbladder followed by the liver and kidney. Radioactivity decreased gradually with time in all organs. The radioactive distribution in the mice organs also showed that the highest %ID/g was in the gallbladder followed by the kidney and liver, and decreased gradually with time. The radioactivity in the gastric system reached its highest level at 60?min. Finally, our study showed the metabolic clearance of 125I-irisin is achieved primarily through the hepatobiliary and renal system and provided the basis for the clinical application of irisin.
Lv J, Pan Y, Li X et al., Horm Metab Res. 2015 Mar 10. [Epub ahead of print]
Context: Polycystic ovary syndrome (PCOS) is an insulin resistance (IR) state, like obesity and type 2 diabetes mellitus (T2DM). Although previous studies have suggested a correlation between irisin and the metabolic parameters associated with obesity and T2DM, the results have been inconsistent.
Objective: Our objective was to 1) determine circulating irisin levels in women with PCOS and control subjects, 2) examine the relationship of irisin and conventional markers of insulin resistance, and 3) examine irisin changes with interventions modulating IR in PCOS women.
Patients and Design: This study was comprised of a series of cross-sectional and interventional studies of 178 PCOS and 123 healthy women from the general population and outpatients of the Internal Medicine Department at the Second Affiliated Hospital, Chongqing Medical University, China. 47 women with PCOS were randomly assigned to 6 months of oral metformin (850 mg bid). OGTT and EHC were performed to assess glucose tolerance and insulin sensitivity. Outcome measures were IR (AUC Insulin and M values) on an OGTT and EHC, irisin levels, and metabolic markers.
Results: Circulating irisin was significantly higher in both overweight/obese (BMI?25 kg/ m2) and PCOS women (P<0.01). Circulating irisin levels correlated with BMI, WHR, FAT%, TG, TC, LDL-C, AUC Insulin, HOMA2-IR, M values and FAI. During EHC, short-term hyperinsulinemia exhibited an inhibitory effect on irisin levels. After 6 months of metformin treatment, there was a significant decrease in circulating irisin in PCOS women following improved IR.
Conclusions: These data suggest that irisin may be a useful marker of IR in PCOS women.
Li M, Yang M, Zhou X, et al., J Clin Endocrinol Metab. 2015 Feb 12:jc20142544. [Epub ahead of print]
BACKGROUND: although there is evidence of correlation between irisin and osteoporotic fractures, previous studies have not elucidated the relationship between irisin and either lean or fat mass. The main aim of this study is to investigate the relationship between irisin and body composition in postmenopausal women with osteoporosis and the impact of irisin levels on fragility vertebral fractures.
METHODS: In this cross-sectional study, 36 overweight subjects affected by at least one vertebral osteoporotic fracture confirmed by a X-ray vertebral morphometry and 36 overweight non-osteoporotic subjects were enrolled. Serum irisin levels were measured using an irisin competitive ELISA. We evaluated lumbar spine and hip BMD and body composition using dual energy X-ray absorptiometry. To measure and monitor daily physical activity, each subject wore an armband for approximately 72 hours.
RESULTS: No significant correlations were found between irisin and BMD at any site and between irisin with either lean or fat mass. Serum levels ofirisin were not correlated with the daily physical activity. Serum irisin levels were lower in subjects with previous osteoporotic fractures than in controls (p= 0.032) and the difference in irisin levels remained significant after adjustment for creatinine (p=0.037), vitamin D (p=0.046), lean mass (p=0.02), lumbar BMD (p=0.023) and femoral BMD (p=0.032).
CONCLUSION: our data confirm an inverse correlation between irisin levels and vertebral fragility fractures but no significant correlation was found with BMD or lean mass. Irisin may play a protective role on bone health independent of BMD but further studies are needed to clarify the relationship between irisin and bone metabolism. This article is protected by copyright. All rights reserved.
Palermo A, Strollo R, Maddaloni E et al., Clin Endocrinol (Oxf). 2014 Nov 17. doi: 10.1111/cen.12672
Irisin is a newly discovered myokine that links exercise with metabolic homeostasis. It is involved in modest weight loss and improves glucose intolerance. However, the direct effects and mechanisms of irisin on vascular endothelial cells (ECs) are not fully understood. In the current study, we demonstrated that irisin promoted Human Umbilical Vein Endothelial Cell (HUVEC) proliferation. It was further demonstrated that this pro-proliferation effect was mediated by irisin-induced activation of extracellular signal-related kinase (ERK) signaling pathways. Inhibition of ERK signaling with U0126 decreased the pro-proliferation effect of irisin on HUVECs. It was also demonstrated that irisin reduced high glucose-induced apoptosis by up-regulating Bcl-2 expression and down-regulating Bax, Caspase-9 and Caspase-3 expression. In summary, these results suggested that irisin plays a novel role in sustaining endothelial homeostasis by promoting HUVEC proliferation via the ERK signaling pathway and protects the cell from high glucose-induced apoptosis by regulating Bcl-2,Bax and Caspase expression.
Song H, Wu F, Zhang Y et al., PLoS One. 2014 Oct 22;9(10):e110273. doi: 10.1371/journal.pone.0110273. eCollection 2014
Ursolic acid (UA), a type of pentacyclic triterpenoid carboxylic acid purified from natural plants, can promote skeletal muscle development. We measured the effect of resistance training (RT) with/without UA on skeletal muscle development and related factors in men. Sixteen healthy male participants (age, 29.37±5.14 years; body mass index=27.13±2.16 kg/m2) were randomly assigned to RT (n=7) or RT with UA (RT+UA, n=9) groups. Both groups completed 8 weeks of intervention consisting of 5 sets of 26 exercises, with 10?15 repetitions at 60-80% of 1 repetition maximum and a 60?90-s rest interval between sets, performed 6 times/week. UA or placebo was orally ingested as 1 capsule 3 times/day for 8 weeks. The following factors were measured pre-and post-intervention: body composition, insulin, insulin-like growth factor-1 (IGF-1), irisin, and skeletal muscle strength. Body fat percentage was significantly decreased (p£¼0.001) in the RT+UA group, despite body weight, body mass index, lean body mass, glucose, and insulin levels remaining unchanged. IGF-1 and irisin were significantly increased compared with baseline levels in the RT+UA group (p£¼0.05). Maximal right and left extension (p£¼0.01), right flexion (p£¼0.05), and left flexion (p£¼0.001) were significantly increased compared with baseline levels in the RT+UA group. These findings suggest that UA-induced elevation of serum irisin may be useful as an agent for the enhancement of skeletal muscle strength during RT.
Hyun Seok Bang, Dae Yun Seo, Yong Min Chung et al., Korean J Physiol Pharmacol. 2014 Oct;18(5):441-446
This study investigated the regulation of thermogenic capacity in classical BAT and SC WAT, and how it affects whole-body energy expenditure in sedentary (Sed) and endurance-trained (Ex) rats fed ad libitum either low-fat (LF) or high-fat (HF) diets. Analysis of tissue mass, PGC-1? and UCP-1 content, presence of multilocular adipocytes, and palmitate oxidation revealed that HF diet increased the thermogenic capacity of the interscapular and aortic brown adipose tissues, while exercise markedly suppressed it. Conversely, exercise induced browning of the subcutaneous inguinal (SC Ing) WAT. This effect was attenuated by HF diet. Endurance training neither affected skeletal muscle FNDC5 content nor circulatingirisin, but it increased FNDC5 content in SC Ing WAT. This suggests that locally produced FNDC5 rather than circulating irisin mediated the exercise-induced browning effect on this fat tissue. Importantly, despite reducing the thermogenic capacity of classical BAT, exercise increased whole-body energy expenditure during the dark cycle. Therefore, browning of SC WAT, likely exerted a compensatory effect and raised whole-body energy expenditure in endurance-trained rats. Based on these novel findings, we propose that exercise-induced browning of the SC WAT provides an alternative mechanism that reduces thermogenic capacity in core areas and increases it in peripheral body regions. This could allow the organism to adjust its metabolic rate to accommodate diet-induced thermogenesis while simultaneously coping with the stress of chronically increased heat production through exercise.
Wu MV, Bikopoulos G, Hung S, Ceddia RB, J Biol Chem. 2014 Oct 25. pii: jbc.M114.591008
BACKGROUND: Skeletal muscles produce irisin. Growing controversy exists on the association between this myokine and chronic disease risk. On the basis of the potential protective effects that irisin could exert on both vascular function and skeletal muscle mass, we hypothesized that an elevated level of this molecule may contribute to successful aging.
METHODS: Serum irisin levels were measured using enzyme-linked immunosorbent assay in disease-free centenarians, young healthy controls, and patients with precocious acute myocardial infarction.
RESULTS: We found the highest levels of serum irisin in disease-free centenarians (35.3 ± 5.5 ng/mL) compared with young healthy controls (20.7 ± 6.3 ng/mL) and especially with young patients with acute myocardial infarction (15.1 ± 5.4 ng/mL).
CONCLUSIONS: Our study demonstrates that healthy centenarians are characterized by increased serum irisin levels, whereas levels of this molecule were found to be significantly lower in young patients with myocardial infarction. Our findings may prompt further research into the role played by irisin not only in vascular disorders but also in life span modulation.
Emanuele E, Minoretti P, Pareja-Galeano H et al., Am J Med. 2014 Sep;127(9):888-90. doi: 10.1016/j.amjmed.2014.04.025
Objective: A recently discovered myokine, irisin, may have an important role in energy metabolism. This study aimed to evaluate the relationship between this hormone and the lipid profile of patients with metabolic syndrome (MetS) following a hypocaloric diet.
Design: Ninety-three Caucasian adults (52 men/41 women) diagnosed with MetS followed an 8-week-long energy-restricted programme (?30% of the energy requirements). Anthropometric measurements, biochemical markers and plasma irisin levels were analysed before and after the nutritional intervention.
Results: Global plasma irisin levels were significantly reduced at the end of the study (?72·0 ± 100·9 ng/ml, P < 0·001) accompanying the weight loss (?6·9%). The depletion of irisin significantly correlated with changes in some atherogenic-related variables: total cholesterol (B = 0·106, P = 0·018), total cholesterol/high-density lipoprotein cholesterol ratio (B = 0·002, P = 0·036), low-density lipoprotein cholesterol (B = 0·085, P = 0·037) and apolipoprotein B (B = 0·052, P = 0·002), independently of changes in body weight.
Conclusions: An association between the reduction in plasma irisin levels and the depletion of important lipid metabolism biomarkers was observed in patients with MetS undergoing an energy-restricted programme.
Rocio de la Iglesia, Patricia Lopez-Legarrea, Ana B. Crujeiras et al., Clin Endocrinol. 2014;81(2):306-311
Background: Peripheral action of irisin improves glucose homeostasis and increases energy expenditure, with no data on a central role of irisin in metabolism. These studies sought to examine (1) presence of irisin in human cerebrospinal fluid (CSF) and banked human hypothalamic tissue, (2) serum irisin in maternal subjects across varying adiposities with or without gestational diabetes (GDM), and (3) their respective neonate offspring.
Methods: CSF, serum and neonatal cord serum were collected from 91 pregnant women with and without GDM attending for an elective Caesarean section (BMI: 37.7±7.6 Kg/m2; age: 32±8.3 years). Irisin was assessed by ELISA and correlated with biochemical and anthropometric dxata. Irisin expression was examined in human hypothalamus by immunohistochemical staining.
Results: Serum irisin in pregnant women was significantly lower in non-obese compared to obese and GDM subjects, after adjusting for BMI, lipids and glucose. Irisin was present in neonatal cord serum (237±8ng/ml) and maternal CSF (32±1.5ng/ml). CSF irisin correlated positively with serum irisin levels from non-obese and obese pregnant women (p<0.01), with CSF irisin significantly raised in GDM subjects (p<0.05). Irisin was present in human hypothalamic sections in the paraventricular neurons, co-localized with neuropeptideY.
Conclusions: Irisin was detectable in CSF and in paraventricular neurons. Maternal serum irisin was lower in non-obese pregnant women after adjusting for BMI and a number of metabolic parameters. These studies indicate that irisin may have a central role in metabolism in addition to the known peripheral role. Further studies investigating the central action of irisin in human metabolic disease are required.
Piya MK, Harte AL, Sivakumar K et al., Am J Physiol Endocrinol Metab. 2014 Jan 7. [Epub ahead of print]
OBJECTIVE: Irisin has recently been introduced as a novel an exercise-inducible myokine which improves glucose metabolism in mice. However, regulation of circulating irisin in gestational diabetes mellitus (GDM) and in the peripartal period has not been assessed so far.
METHODS: Circulating irisin was quantified in 74 GDM patients and in 74 healthy, pregnant, gestational age-matched controls. In a subset of these patients (44 GDM, 41 controls), postpartum follow-up data were also available. In a second study population of 40 healthy women with singleton pregnancies undergoing elective Cesarean section, irisin was assessed in maternal serum before and within 24h after delivery, as well as in umbilical cord blood and in placental tissue.
RESULTS: In the first study population, median [interquartile range] irisin levels were significantly higher in GDM patients as compared to controls after delivery (previous GDM: 446.3 [146.9]?g/l; controls: 378.0 [111.4]?g/l) but not during pregnancy (GDM: 482.1 [132.1]?g/l; controls: 466.6 [178.0]?g/l). Interestingly, fasting insulin (FI) was independently and positively associated with serum irisin in multivariate analysis during pregnancy. In agreement with these findings, relative changes (ratio) of FI independently and positively predicted relative changes of irisin (ratio) in the second study population.
CONCLUSIONS: The myokine irisin is independently associated with FI in pregnancy. The physiological significance of these findings needs to be assessed in future experiments.
This publication used an Irisin kit from Phoenix Pharmaceuticals.Ebert T, Stepan H, Schrey S et al., Cytokine. 2013 Dec 16. pii: S1043-4666(13)00759-X. doi: 10.1016/j.cyto.2013.11.009. [Epub ahead of print]
Irisin converts white adipose tissue (WAT) into brown adipose tissue (BAT), as regulated by energy expenditure. The relationship between irisin concentrations after exercise in rats compared humans after exercise remains controversial. We therefore: 1) measured irisin expression in cardiac and skeletal muscle, liver, kidney, peripheral nerve sheath and skin tissues, as also serum irisin level in 10 week-old rats without exercise, and 2) measured tissue supernatant irisin levels in cardiac and skeletal muscle, and in response to exercise in young and old rats to establishing which tissues produced most irisin. Young (12 months) and old rats (24 months) with or without 10min exercise (water floating) and healthy 10 week-old Sprague-Dawley rats without exercise were used. Irisin was absent in sections of skeletal muscle of unexercised rats, the only part being stained being the perimysium. In contrast, cardiac muscle tissue, peripheral myelin sheath, liver, kidneys, and skin dermis and hypodermis were strongly immunoreactivity. No irisin was seen in skeletal muscle of unexercised young and old rats, but a slight amount was detected after exercise. Strong immunoreactivity occurred in cardiac muscle of young and old rats with or without exercise, notably in pericardial connective tissue. Serum irisin increased after exercise, being higher in younger than older rats. Irisin in tissue supernatants (cardiac and skeletal muscle) was high with or without exercise. High supernatant irisin could come with connective tissues around skeletal muscle, especially nerve sheaths located within it. Skeletal muscle is probably not an irisin source.
Aydin S, Kuloglu T, Aydin S et al., Peptides. 2013 Dec 14. pii: S0196-9781(13)00410-5. doi: 10.1016/j.peptides.2013.11.024. [Epub ahead of print]
Irisin was identified as a myokine secreted by contracting skeletal muscle, possibly mediating some of exercise health benefits via "browning" of white adipose tissue. However, a controversy exists concerning irisin origin, regulation&function in humans. Thus, we have explored Fndc5/irisin in 2 clinical studies: (i) a cross-sectional study (effects of type-2-diabetes (T2D) in drug-naïve men); (ii) an intervention study (exercise effects in sedentary, overweight/obese individuals). Methods. Glucose tolerance and insulin sensitivity were assessed. VO2max and muscle strength were measured before and after training. Body composition (MRI), muscle&liver fat content (1H-MRS) and in vivo muscle metabolism (32P-MRS) were determined. Skeletal muscle and subcutaneous abdominal adipose tissue samples were taken in the fasted state, during euglycemic hyperinsulinemia (adipose tissue) and before/after exercise training (muscle). Results. Muscle Fndc5 mRNA was increased in prediabetes but not T2D. Fndc5/irisin in adipose tissue/plasma were reduced in T2D by 40% and 50%, respectively. Opposite to that, T2D-derived myotubes expressed/secreted the highest levels of Fndc5/irisin. Neither hyperinsulinemia (adipose tissue/plasma) nor exercise (muscle/plasma) affected Fndc5/irisin in vivo. Circulating irisin was positively associated with muscle mass, strength and metabolism and negatively with fasting glycemia. Glucose and palmitate decreased Fndc5 mRNA in myotubes in vitro. Conclusions. Distinct patterns of Fndc5/irisin in muscle, adipose tissue nad circulation and concordant in vivo down-regulation in T2D indicate that irisin might distinguish metabolic health and disease. Moreover, Fndc5/irisin was discordantly regulated in diabetic muscle and myotubes in vitro, suggesting that whole-body factors, such as glucose and fatty acids, might be important for irisin regulation. Exercise did not affect Fndc5/irisin. However, irisin was positively linked to muscle mass, strength and metabolism, pointing to common regulatory factors and/or the potential for irisin to modify muscle phenotype.
Fig.1 of example #1 . Plasma irisin concentration (A) and Fndc5 gene expression in skeletal muscle (C) and subcutaneous adipose tissue (E) in individuals with obesity, prediabetes and type 2 diabetes. Data are shown as means ±SEM. *p<0.05. ** p<0.01. *** p<0.005
Subset of the 21 plasma samples from the study were measured with a more recently developed recombinant irisin EIA kit (#EK-067-29). This assay provided authors with identical results, but revealed that previously measured plasma concentrations might be underestimated by approximately 25 fold. Figure 3 of example #1. Fndc5/irisin Subset of the differentiated human primary muscles originating from lean, obese, prediabetes and tupe 2 diabetic individuals. (A) Fndc5 mRNA in differentiated human primary muscle cells number of samples from experiment 2. (B) Irisin in the conditioned media from human primary muscle cells. Data are shown as means ± SEM. *p<0.05.
Kurdiova T, Balaz M, Vician M et al., J Physiol. 2013 Dec 2. [Epub ahead of print]
Objective: A recently discovered myokine, irisin, may have an important role in energy metabolism. This study aimed to evaluate the relationship between this hormone and the lipid profile of Metabolic Syndrome (MetS) patients following a hypocaloric diet.
Design: Ninety-three Caucasian adults (52 men/41 women) diagnosed with MetS followed an 8-week-long energy restricted programme (-30% of the energy requirements). Anthropometric measurements, biochemical markers and plasma irisin levels were analysed before and after the nutritional intervention.
Results: Global plasma irisin levels were significantly reduced at the end of the study (-72.0 ± 100.9 ng/mL, p < 0.001) accompanying the weight loss (-6.9%). The depletion of irisin significantly correlated with changes in some atherogenic-related variables: total cholesterol (B = 0.106, p = 0.018), total cholesterol/high density lipoprotein-cholesterol ratio (B = 0.002, p = 0.036), low density lipoprotein-cholesterol (B = 0.085, p = 0.037) and apolipoprotein B (B = 0.052, p = 0.002), independently of changes in body weight. Conclusions An association between the reduction of plasma irisin levels and the depletion of important lipid metabolism biomarkers was observed in patients with MetS undergoing an energy restricted program.
de la Iglesia R, Lopez-Legarrea P, Crujeiras AB, et al., Clin Endocrinol (Oxf). 2013 Dec 11. doi: 10.1111/cen.12383. [Epub ahead of print]
AIMS: Chronic kidney disease (CKD) secondary to type 2 diabetes mellitus (T2DM) is associated with multifaceted energy dysmetabolism. We aim to study the relationship between renal function, body composition and irisin, the recently identified myokine which is involved in energy regulation, in T2DM.
METHODS: Circulating irisin and body composition were measured in 365 T2DM subjects across a wide range of renal function.
RESULTS: Circulating irisin was significantly decreased in T2DM with renal insufficiency (77.4 ¡À 13.7ng/ml in T2DM with eGFR ~60ml/min/1.73m2 versus 72.5 ± 14.9ng/ml in those with eGFR<60ml/min/1.73m2, p=0.001) and the reduction in irisin was most pronounced in stage 5 CKD patients. In T2DM with preserved renal function, irisin was correlated with age (r=-0.242, p=0.001) and pulse pressure (r=-0.188, p=0.002). Among those with renal insufficiency, irisin was correlated with BMI (r=0.171, p=0.022), fat mass (r=0.191, p=0.013), percentage of fat mass (r=0.210, p=0.007) and eGFR (r=0.171, p=0.020). Multivariate linear regression models revealed that variations in circulating irisin were mainly attributable to eGFR and age in T2DM with and without renal impairment, respectively.
CONCLUSION: Our observations suggest that the level of circulating irisin may be associated with renal function in T2DM. The role of reduced irisin in energy dysmetabolism in diabetic patients with renal insufficiency deserves further investigation.
Liu JJ, Liu S, Wong MD et al., J Diabetes Complications. 2013 Oct 4. pii: S1056-8727(13)00214-6. doi: 10.1016/j.jdiacomp.2013.09.011. [Epub ahead of print]
Irisin was recently identified as a putative myokine that is induced by exercise. Studies suggest that it is produced by cleavage of the FNDC5 (fibronectin domain-containing protein 5) receptor; irisin corresponds to the extracellular receptor ectodomain. Data suggesting that irisin stimulates white to brown fat conversion has led to the hypothesis that it does so by binding an unknown receptor, thus functioning as a myokine. As brown fat promotes energy dissipation, myokines that elicit the transformation of white to brown fat have potentially profound benefits in the treatment of obesity and metabolic disorders. Understanding the molecular basis for such exercise-induced phenomena is thus, of considerable interest. Moreover, FNDC5-like receptors are highly conserved and have been shown to be critical for neuronal development. However, the structural and molecular mechanisms utilized by these proteins are currently unknown. Here we describe the crystal structure and biochemical characterization of the FNDC5 ectodomain, corresponding to the irisin myokine. The 2.28 Angstrom structure shows that irisin consists of an N-terminal fibronectin III(FNIII)-like domain attached to a flexible C-terminal tail. Strikingly, the FNIII-like domain forms a continuous intersubunit ?-sheet dimer, previously unobserved for any FNIII protein. Biochemical data confirm that irisin is a dimer and that dimerization is unaffected by glycosylation. This finding suggests a possible mechanism for receptor activation by the irisin domain as a preformed myokine dimer ligand or as a paracrine or autocrine dimerization module on FNDC5-like receptors.
Schumacher MA, Chinnam N, Ohashi T et al., J Biol Chem. 2013 Oct 10. 1074/jbc.M113.516641 [Epub ahead of print]
The number and activity of brown adipocytes are linked to the ability of mammals to resist body fat accumulation. In some conditions, certain white adipose tissue (WAT) depots are readily convertible to a \'\'brown-like\'\' statewhich is associated with weight loss. Irisin, a newly identified hormone, is secreted by skeletal muscles into circulation and promotes WAT \"browning\" with unknown mechanisms. In the current study, we demonstrated in mice that recombinant irisin decreased the bodyweight and improved glucose homeostasis. We further showed that irisin upregulated UCP-1 (a regulator of thermogenic capability of brown fat) expression. This effect was possibly mediated by irisin induced phosphorylation of p38 MAPK and ERK signaling pathway. Inhibition of the p38 MAPK by SB203580 and ERK by U0126 abolished the upregulatory effect of irisin on UCP-1. In addition, irisin also promoted the expression of betatrophin, another newly identified hormone that promotes pancreatic beta cell proliferation and improves glucose tolerance. In summary, our data suggest that irisin can potentially prevent obesity and associated type 2 diabetes by stimulating expression of WAT browning-specific genes via p38 MAPK and ERK pathway.
Zhang Y, Li R, Meng Y, Li S, et al., Diabetes. 2013 Oct 22. [Epub ahead of print]
The purpose of this study was to ascertain (1) whether human saliva contains irisin and whether its level correlates with serum irisin concentration, (2) whether salivary glands, eccrine glands and sebaceous glands in human skin produce irisin, (3) how the changes in saliva and serum irisin concentrations after the Turkish bath at 47±3°C compare with the changes caused by moderate exercise in obese and normal weight subjects. Seven obese male subjects and seven normal weight subjects were enrolled for Turkish bath. Seven obese male subjects and seven normal weight subjects were also enrolled for moderate outdoor exercise, and thirteen male normal weight subjects neither exercised nor showered at the Turkish bath. From each participant, 1.5ml of saliva and 5ml blood were collected simultaneously before and after the moderate exercise and Turkish bath. Salivary glands and eccrine and sebaceous glands in the skin were screened immunohistochemically for irisin while serum and saliva irisin were measured with an ELISA. Submandibular glands, eccrine glands and sebaceous glands in the human skin showed strong irisin immunoreactivity. Human saliva contained irisin and its level was significantly higher than the serum levels in both obese and normal weight subjects. However, irisin concentrations were more markedly increased in both saliva and serum samples from subjects who had showered at a Turkish bath than in obese subjects who had exercised or in normal weight subjects. Human submandibular glands, eccrine sweat glands and sebaceous glands synthesize irisin.
Aydin S, Kuloglu T, Yilmaz M, et al., Peptides. 2013 Oct 1. pii: S0196-9781(13)00324-0. doi: 10.1016/j.peptides.2013.09.011. [Epub ahead of print]
OBJECTIVE: Skeletal muscle is considered to be an endocrine organ that secretes a number of myokines including follistatin, myostatin, activin A and the newly identified irisin. Irisin\'s biology and function exhibits similarities with the functions of the follistatin-myostatin-activin A axis. It remains unknown whether there is any interplay among these molecules. The aim of this study is to examine potential associations of irisin with the follistatin, myostatin and activin A axis.
MATERIAL-METHODS: Two observational studies were performed to evaluate the associations of irisin with the other three peptides. Study A included 150 healthy males aged 18.48 ±0.16 years with Body Mass Index (BMI) 23.18± 3.75 kg/m². Fasting serum samples were used to measure the levels of the molecules of interest. Study B included 14 morbidly obese individuals, candidates for bariatric surgery, aged 53.14±8.93 years with BMI 50.18±10.63 kg/m². Blood samples were obtained after an overnight fast. Eight out of the fourteen participants consented to an optional thigh biopsy during their bariatric surgery. Using the above blood and tissue samples, we measured circulating levels and muscle mRNA of irisin, follistatin, myostatin and activin A.
RESULTS: We report that FNDC5 mRNA in muscle is positively correlated with follistatin mRNA expression in morbidly obese subjects (rho=0.93, p<0.001). We also found that circulating irisin is positively correlated with follistatin circulating levels among lean subjects (rho=0.17, p=0.05) while this association was suggestive among the obese (rho=0.56, p=0.07).
CONCLUSION: The newly identified myokine irisin may be positively associated with follistatin at both the mRNA and circulating protein level.
Vamvini MT, Aronis KN, Panagiotou G, et al., Eur J Endocrinol. 2013 Sep 23. [Epub ahead of print]
Irisin, a newly discovered, PGC-1α dependent myokine, has recently been shown to increase in circulation in response to sprint exercise. This study examined the effect of prolonged exercise on irisin concentrations in young men (n=7) as well as in young women (n=5) during different stages of the menstrual cycle. Seven young men completed 90 min of treadmill exercise at 60% of VO2max and a resting control trial. Five women completed the same exercise protocol in two different trials: during the early follicular phase and mid-luteal phase of the menstrual cycle. Blood samples were collected and analyzed for irisin concentrations immediately before exercise, at 54 and 90 min of exercise, and at 20 min of recovery (R20). Findings revealed that by 54 min of a 90 min treadmill exercise protocol at 60% of VO2max, irisin concentrations significantly increased 20.4% in young men and 20.3% as well as 24.6% in young women during the early follicular and mid-luteal phases of the menstrual cycle, respectively. However, by 90 min of exercise as well as R20, irisin concentrations were no longer elevated. Stage of the menstrual cycle did not affect responses in young women. Findings indicate that prolonged aerobic exercise produces a transient increase in irisin concentrations during the first hour of exercise for both genders and suggest that this form of moderate exercise may be helpful in improving fat metabolism.
Kraemer RR, Shockett P, Webb ND, et al., Horm Metab Res. 2013 Sep 23. [Epub ahead of print]
Context: Irisin, a recently identified hormone, has been proposed to regulate energy homeostasis and obesity in mice. Whether irisin levels are associated with risk of the metabolic syndrome (MetS), cardiometabolic variables, and cardiovascular disease (CVD) risk in humans remains unknown.
Objective: To assess the associations between baseline serum irisin levels and MetS, cardiometabolic variables, and CVD risk.
Design, Setting, and Subjects: Comparative cross-sectional evaluation of baseline circulating levels of the novel hormone irisin, and the established adipokine adiponectin with MetS, cardiometabolic variables, and CVD risk in a sample of 151 subjects.
Results:Baseline irisin levels were significantly higher in subjects with MetS compared to subjects without MetS. Irisin was associated negatively with adiponectin (r= -0.4, p<0.001) and positively with BMI (r=0.22, p=0.008), systolic (r=0.17, p=0.04), diastolic blood pressure (r=0.27, p=0.001), fasting glucose (r=0.25, p=0.002), triglycerides (r=0.25, p=0.003), and HOMA-IR (r=0.33, p<0.001). After adjustment for potential confounders, including BMI, subjects in the highest tertile of irisin levels were more likely to have MetS (OR 9.44, 95%CI 2.66-33.44), elevated fasting blood glucose (OR 5.80, 95%CI 1.72-19.60), high triglycerides (OR 3.89, 95%CI 1.16-13.03), and low HDL-cholesterol (OR 3.30, 95%CI 1.18-9.20). Irisin was independently associated with HOMA-IR and general Framingham risk profile in multiple linear regression analyses after adjustment for confounders. Adiponectin demonstrated the expected associations with outcomes.
Conclusions: Irisin is associated with increased risk of MetS, cardiometabolic variables, and CVD in humans, indicating either increased secretion by adipose/muscle tissue and/or a compensatory increase of irisin to overcome an underlying irisin resistance in these subjects.
This publication used a Irisin kit from Phoenix PharmaceuticalsPark KH, Zaichenko L, Brinkoetter M et al., J Clin Endocrinol Metab. 2013 Sep 20. [Epub ahead of print]
Patients with chronic kidney disease have abnormal energy expenditure and metabolism. The mechanisms underlying altered energy expenditure in uremia are unknown and remain to be elucidated. Irisin is a peroxisome proliferator-activated receptor c coactivator 1-a–dependent myokine, and it increases energy expenditure in the absence of changes in food intake or activity. We hypothesize that chronic kidney disease patients have altered irisin levels. We measured resting irisin levels in 38 patients with stage 5 chronic kidney disease and in 19 age- and sex-matched normal subjects. Plasma irisin levels were significantly decreased in chronic kidney disease patients (58.59%; 95% CI 47.9%–69.2%, p,0.0001). The decrease in irisin levels was inversely correlated with the levels of blood urea nitrogen and creatinine. Further association analysis revealed that irisin level is independently associated with high-density lipoprotein cholesterol level. Our results suggest that chronic kidney disease patients have lower than normal irisin levels at rest. Furthermore, irisin may play a major role in affecting high-density lipoprotein cholesterol levels and abnormal energy expenditure in chronic kidney disease patients.
Wen MS, Wang CY, Lin SL, Hung KC. Decrease in irisin in patients with chronic kidney disease. PLoS ONE. 2013;8(5):e64025
OBJECTIVE: There is debate as to whether fibronectin type III domain containing 5 (FNDC5) and its protein product irisin are therapeutic targets for obesity-associated maladies. Thus, we sought to examine FNDC5 mRNA within skeletal muscle of obese/diabetic-prone Otsuka Long-Evans Tokushima Fatty (OLETF) rats versus lean/healthy Long Evans Tokushima Otsuka (LETO) rats. We hypothesized that FNDC5 expression would be greater in obese (OLETF) versus lean (LETO) animals.
MATERIALS/METHODS: Triceps muscle of 30-32week old OLETF and LETO rats were assayed for FNDC5 and PGC1? mRNA levels. Body composition and circulating biomarkers of the OLETF and LETO rats were also correlated with skeletal muscle FNDC5 mRNA expression patterns in order to examine potential relationships that may exist.
RESULTS: OLETF rats exhibited twice the amount of triceps FNDC5 mRNA compared to LETO rats (p<0.01). Significant positive correlations existed between triceps muscle FNDC5 mRNA expression patterns versus fat mass (r=0.70, p=0.008), as well as plasma leptin (r=0.82, p<0.001). PGC1? mRNA levels were also highly correlated with FNDC5 mRNA (r=0.85, p<0.001). In subsequent culture experiments, low and high physiological doses of leptin had no effect on PGC1? mRNA or FNDC5 mRNA levels in C2C12 myotubes. Paradoxically, circulating irisin concentrations tended to be higher in a second cohort of LETO versus OLETF rats (p=0.085).
CONCLUSION: These results reveal a positive association between total body adiposity and skeletal muscle FNDC5 gene expression. Of interest, circulating irisin levels tended to be lower in OLETF rats. Further research is needed to examine whether other adipose tissue-derived factors up-regulate FNDC5 transcription and/or inhibit irisin biosynthesis from FNDC5.
This publication used an Irisin kit from Phoenix Pharmaceuticals.
Roberts MD et al, Metabolism. 2013 Mar 13. pii: S0026-0495(13)00044-9. doi: 10.1016/j.metabol.2013.02.002. [Epub ahead of print]
Irisin is a recently identified myokine secreted from the muscle in response to exercise. In the rats and mice, immunohistochemical studies with an antiserum against irisin peptide fragment (42-112), revealed that irisin-immunoreactivity (irIRN) was detected in three types of cells; namely, skeletal muscle cells, cardiomyocytes, and Purkinje cells of the cerebellum. Tissue sections processed with irisin antiserum pre-absorbed with the irisin peptide(42-112) (1 ug/ml) showed no immunoreactivity. Cerebellar Purkinje cells were also immunolabeled with an antiserum against fibronectin type II domain containing 5 (FNDC5), the precursor protein of irisin. Double-labeling of cerebellar sections with irisin antiserum and glutamate decarboxylase (GAD) antibody showed that nearly all irIRN Purkinje cells were GAD-positive. Injection of the fluorescence tracer Fluorogold into the vestibular nucleus of the rat medulla retrogradely labeled a population of Purkinje cells, some of which were also irIRN. Our results provide the first evidence of expression of irIRN in the rodent skeletal and cardiac muscle, and in the brain where it is present in GAD-positive Purkinje cells of the cerebellum. Our findings together with reports by others led us to hypothesize a novel neural pathway, which originates from cerebellum Purkinje cells, via several intermediary synapses in the medulla and spinal cord, and regulates adipocyte metabolism.
This publication used a Irisin and FNDC5 antibody from Phoenix Pharmaceuticals.
Dun SL et al., Neuroscience 240 (2013) 155-162. June 14, 2013
AIMSHYPOTHESIS: Recently, the novel myokine irisin was described to drive adipose tissue \'browning\', to increase energy expenditure, and to improve obesity and insulin resistance in high fat-fed mice. Here, we assessed whether common single nucleotide polymorphisms (SNPs) in the FNDC5 locus, encoding the irisin precursor, contribute to human prediabetic phenotypes (overweight, glucose intolerance, insulin resistance, impaired insulin release).
METHODS: A population of 1,976 individuals was characterized by oral glucose tolerance tests and genotyped for FNDC5 tagging SNPs. Subgroups underwent hyperinsulinaemic-euglycaemic clamps, magnetic resonance imaging/spectroscopy, and intravenous glucose tolerance tests. From 37 young and 14 elderly participants recruited in two different centres, muscle biopsies were obtained for the preparation of human myotube cultures.
RESULTS: After appropriate adjustment and Bonferroni correction for the number of tested variants, SNPs rs16835198 and rs726344 were associated with in vivo measures of insulin sensitivity. Via interrogation of publicly available data from the Meta-Analyses of Glucose and Insulin-related traits Consortium, rs726344\'s effect on insulin sensitivity was replicated. Moreover, novel data from human myotubes revealed a negative association between FNDC5 expression and appropriately adjusted in vivo measures of insulin sensitivity in young donors. This finding was replicated in myotubes from elderly men.
CONCLUSIONS & INTERPRETATION: This study provides evidence that the FNDC5 gene, encoding the novel myokine irisin, determines insulin sensitivity in humans. Our gene expression data point to an unexpected insulin-desensitizing effect of irisin.
Staiger H, Böhm A, Scheler M et al, PLoS One. 2013 Apr 25;8(4):e61903. doi: 10.1371/journal.pone.0061903. Print 2013
AIMS: Irisin is a novel myokine secreted in response to PPAR-? co-activator-1? (PGC-1?) activation. Earlier studies suggested that PGC-1α expression and activity were lower in myocytes in type 2 diabetes mellitus (T2DM). Therefore, we hypothesize that circulating irisin levels are lower in T2DM patients.
METHODS: In this observational study, we recruited 96 T2DM subjects and 60 non-diabetic control subjects. Among T2DM subjects, 38% were on insulin treatment, 78% were taking statins and 72% were taking renin-angiotensin system antagonists. Circulating irisin was quantified by ELISA and its association with markers of metabolic phenotype was analyzed by Pearson bivariate correlation and multiple linear regression.
RESULTS: Circulating irisin was significantly lower in individuals with T2DM compared with non-diabetic controls (T2DM 204±72ng/ml vs. non-diabetic control 257±24ng/ml, p<0.0001). In non-diabetic subjects, circulating irisin was correlated with age (r=0.398, p<0.01), BMI (r=0.387, p<0.01), total cholesterol (r=0.341, p<0.01), total triglycerides (r=0.299, p<0.05), fasting blood glucose (r=0.430, p<0.01) and diastolic blood pressure (r=0.306, p<0.05). Multiple linear regression model revealed that BMI (?=0.407, p=0.012) and FBG (?=0.315, p=0.034) were associated with irisin in non-diabetic subjects after adjusting for multiple co-variates. However, similar analysis in T2DM subjects didn\'t reveal significant association between circulating irisin and major markers of metabolic phenotype.
CONCLUSIONS: Circulating irisin is lower in T2DM compared with non-diabetic controls. Plasma irisin levels appear to be associated with important metabolic factors in non-diabetic subjects but not in individuals with type 2 diabetes.
Liu JJ, Wong MD, Toy WC et al, J Diabetes Complications. 2013 Apr 22. pii: S1056-8727(13)00057-3. doi: 10.1016/j.jdiacomp.2013.03.002. [Epub ahead of print]
Exercise provides clear beneficial effects for the prevention of numerous diseases. However, many of the molecular events responsible for the curative and protective role of exercise remain elusive. The recent discovery of FNDC5/irisin protein that is liberated by muscle tissue in response to exercise might be an important finding with regard to this unsolved mechanism. The most striking aspect of this myokine is its alleged capacity to drive brown-fat development of white fat and thermogenesis. However, the nature and secretion form of this new protein is controversial. The present study reveals that rat skeletal muscle secretes a 25 kDa form of FNDC5, while the 12 kDa/irisin theoretical peptide was not detected. More importantly, this study is the first to reveal that white adipose tissue (WAT) also secretes FNDC5; hence, it may also behave as an adipokine. Our data using rat adipose tissue explants secretomes proves that visceral adipose tissue (VAT), and especially subcutaneous adipose tissue (SAT), express and secrete FNDC5. We also show that short-term periods of endurance exercise training induced FNDC5 secretion by SAT and VAT. Moreover, we observed that WAT significantly reduced FNDC5 secretion in fasting animals. Interestingly, WAT of obese animals over-secreted this hormone, which might suggest a type of resistance. Because 72% of circulating FNDC5/irisin was previously attributed to muscle secretion, our findings suggest a muscle-adipose tissue crosstalk through a regulatory feedback mechanism.
Roca-Rivada A, Castelao C, Senin LL et al, PLoS One. 2013 Apr 11;8(4):e60563. doi: 10.1371/journal.pone.0060563. Print 2013
OBJECTIVE: In mouse, PGC1-α overexpression in muscle stimulates an increase in expression of FNDC5, a membrane protein that is cleaved and secreted as a newly identified hormone, irisin. One prior study has shown that FNDC5 induces browning of subcutaneous fat in mice and mediates beneficial effects of exercise on metabolism, but a more recent study using gene expression arrays failed to detect a robust increase in FNDC5 mRNA in human muscles from exercising subjects. No prior study has reported on the physiological regulation and role of circulating irisin and FNDC5 in humans.
MATERIALS/METHODS: A. FNDC5 gene expression studies: We first examined tissue distribution of FNDC5 in humans. B. Cross-sectional studies: Predictors of FNDC5 mRNA expression levels were examined in muscle tissues from 18 healthy subjects with a wide range of BMI. Assays were optimized to measure circulating FNDC5 and irisin levels, and their associations with anthropometric and metabolic parameters were analyzed in two cross-sectional studies that examined 117 middle-aged healthy women and 14 obese subjects, respectively. C. Interventional studies: The effect of weight loss on FNDC5 mRNA and/or circulating irisin levels was examined in 14 obese subjects before and after bariatric surgery. The effect of acute and chronic exercise was then assessed in 15 young healthy adults who performed intermittent sprint running sessions over an 8week period.
RESULTS: Tissue arrays demonstrated that in humans, the FNDC5 gene is predominantly expressed in muscle. Circulating irisin was detected in the serum or plasma of all subjects studied, whereas circulating FNDC5 was detected in only a distinct minority of the subjects. Cross-sectional studies revealed that circulating irisin levels were positively correlated with biceps circumference (used as a surrogate marker of muscle mass herein), BMI, glucose, ghrelin, and IGF-1. In contrast, irisin levels were negatively correlated with age, insulin, cholesterol, and adiponectin levels, indicating a possible compensatory role of irisin in metabolic regulation. Multivariate regression analysis revealed that biceps circumference was the strongest predictor of circulating irisin levels underlying the association between irisin and metabolic factors in humans at baseline. Both muscle FNDC5 mRNA levels and circulating irisin levels were significantly downregulated 6months after bariatric surgery. Circulating irisin levels were significantly upregulated 30min after acute exercise and were correlated mainly with ATP levels and secondarily with metabolites related to glycolysis and lipolysis in muscle.
CONCLUSIONS: Similar to mice, the FNDC5 gene is expressed in human muscle. Age and muscle mass are the primary predictors of circulating irisin, with young male athletes having several fold higher irisin levels than middle-aged obese women. Circulating irisin levels increase in response to acute exercise whereas muscle FNDC5 mRNA and circulating irisin levels decrease after surgically induced weight loss in parallel to decrease in body mass. Further studies are needed to study the regulation of irisin levels and its physiological effects in humans and to elucidate the mechanisms underlying these effects.
Huh JY, Panagiotou G, Mougios V, et al, Metabolism. 2012 Sep 24. pii: S0026-0495(12)00332-0. doi: 10.1016/j.metabol.2012.09.002
Background—Exercise induced increase in peroxisome proliferator-activated receptor-γ co-activator-1α (PGC-1α) expression has been shown to increase the expression of the fibronectin type III domain containing 5 (FNDC5) gene and thereby its product, irisin, in mice. Given that exercise intolerance is a hallmark characteristic of heart failure (HF), and since PGC-1? and irisin promote exercise benefits in animals, we hypothesized that expression of these genes relates to aerobic performance in patients with HF. Methods and Results—Systolic HF (LVEF ≤40%) patients underwent cardiopulmonary exercise testing (CPX) to evaluate aerobic performance. High vs. low aerobic performance was assessed using oxygen consumption [peak VO2 (>14mlO2•kg-1•min-1 vs. ≥14 mlO2•kg-1•min-1)] and ventilatory efficiency [VE/VCO2 slope (<34 vs. ≥34)]. Muscle biopsies of the vastus lateralis and real-time polymerase chain reaction were used to quantify muscle gene expression. Twenty-four patients were studied. FNDC5 (5.7±3.5 vs. 3.1±1.2, p<.05) and PGC-1α (9.9±5.9 vs. 4.5±1.9, p<.01) gene expression was greater in the high peak VO2 group; correlation between FNDC5 and PGC-1? was significant (r=0.56, p<.05) only in the high peak VO2 group. Similarly, FNDC5 and PGC-1α gene expression was greater in the high performance group based on lower VE/VCO2 slopes (5.8±3.6 vs. 3.3±1.4, p<.05 and 9.7±6 vs. 5.3±3.4, p<.05); FNDC5 also correlated with PGC-1α (r=0.55, p<.05) only in the low VE/VCO2 slope group. Conclusions—This is the first study to show that FNDC5 expression relates to functional capacity in a human HF population. Lower FNDC5 expression may underlie reduced aerobic performance in HF patients.
Lecker S. et al., CIRCHEARTFAILURE.112.969543, September 20, 2012, doi: 10.1161/CIRCHEARTFAILURE.112.969543
Exercise benefits a variety of organ systems in mammals, and some of the best-recognized effects of exercise on muscle are mediated by the transcriptional co-activator PPAR-gamma co-activator-1 a (PGC1-a). Here we show in mouse that PGC1-a expression in muscle stimulates an increase in expression of FNDC5, a membrane protein that is cleaved and secreted as a newly identified hormone, irisin. Irisin acts on white adipose cells in culture and in vivo to stimulate UCP1 expression and a broad program of brown-fat-like development. Irisin is induced with exercise in mice and humans, and mildly increased irisin levels in the blood cause an increase in energy expenditure in mice with no changes in movement or food intake. This results in improvements in obesity and glucose homeostasis. Irisin could be therapeutic for human metabolic disease and other disorders that are improved with exercise.
Boström et al., Nature. 2012 Jan 11. doi: 10.1038/nature10777. [Epub ahead of print]
The identification of several peroxisomal proteins in the past decade has deepened our understanding of the biology of peroxisomes and their involvement in human disorders. We report the cloning and expression pattern during the mouse development of a cDNA encoding a novel protein, named PeP, and show that its product is imported specifically to the peroxisome matrix in a variety of cell types. We also demonstrate that PeP is imported to the organelle through the PEX5 receptor pathway, which indicates that the C-terminal tripeptide SKI behaves as a type 1 peroxisomal targeting signal (PTS1). PeP expression is tightly regulated, as shown by Northern and in situ hybridization experiments. Thus during embryonic development in the mouse, PeP mRNA is detected almost exclusively in the skeletal muscle, whereas in adult mice, strong expression is also found in the heart and brain. In addition, PeP mRNA accumulation is induced after myoblast differentiation in vitro, when myotube formation is promoted. Sequence analysis reveals that PeP has no significant homology to any known protein, except for a short stretch of amino acids containing the fingerprint of the fibronectin type III superfamily, a domain present in proteins often related to molecular and cellular recognition and binding processes. Thus our data suggest a connection between the function of PeP and murine cell differentiation and development.
Ferrer-martínez A, Ruiz-lozano P, Chien KR. Dev Dyn. 2002;224(2):154-67.
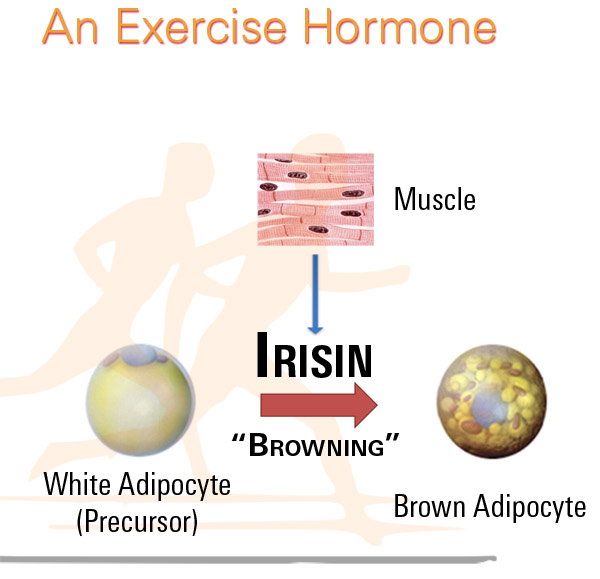
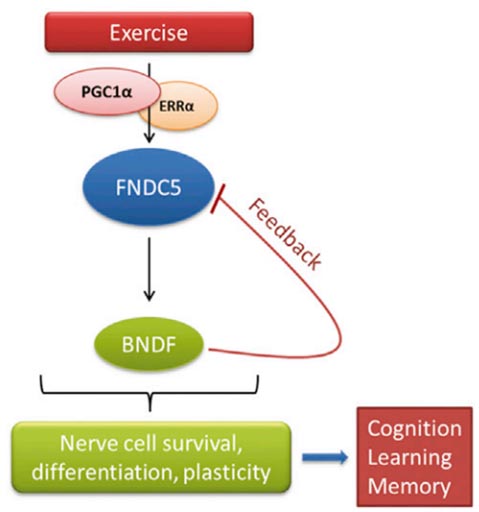
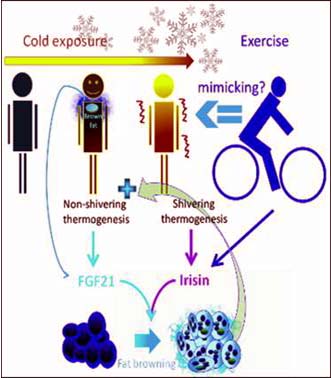
Nonshivering cold exposure increases FGF21, which may be a brown adipokine.
• Irisin and/or FGF21 upregulates brown-fat-like program in human adipocytes
• Exercise may be a shivering mimic exemplifying muscle-fat thermogenic crosstalk.
Figure from: Cell Metabolism, Volume 19, Issue 2, 302-309.
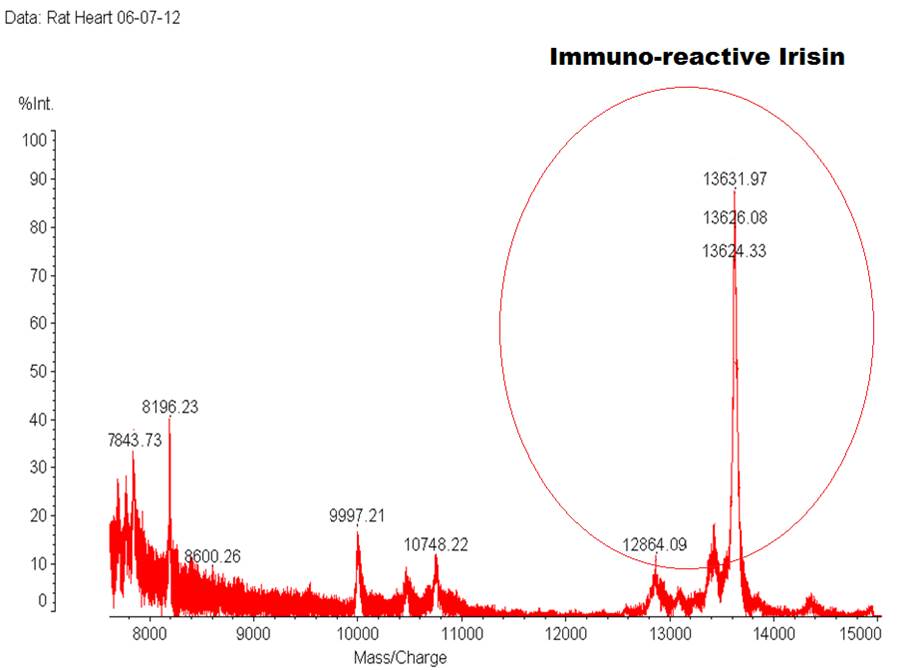
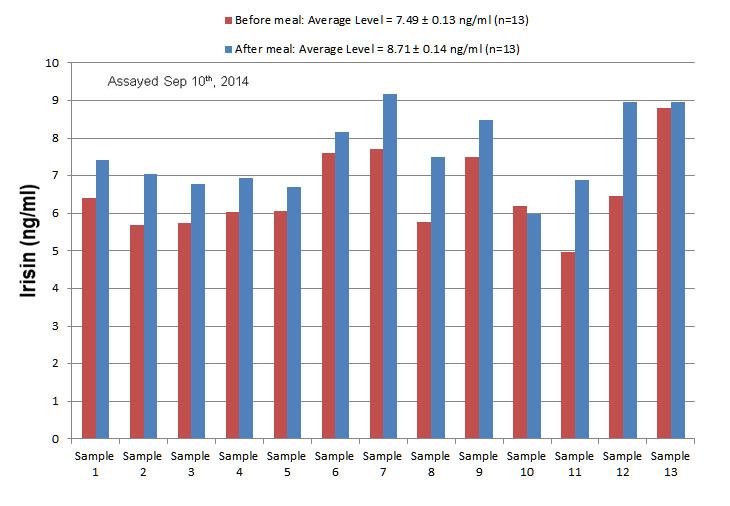
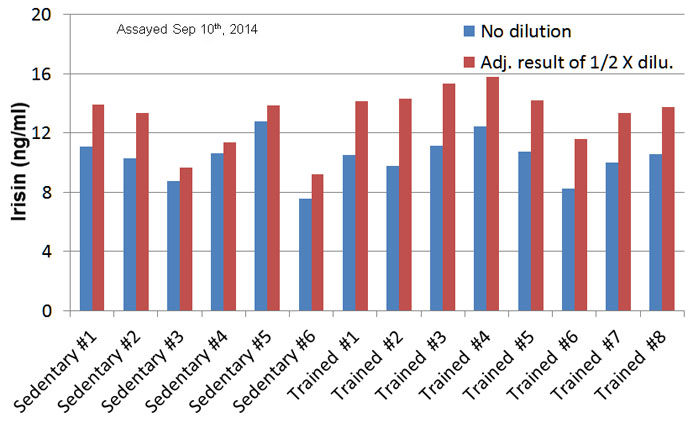
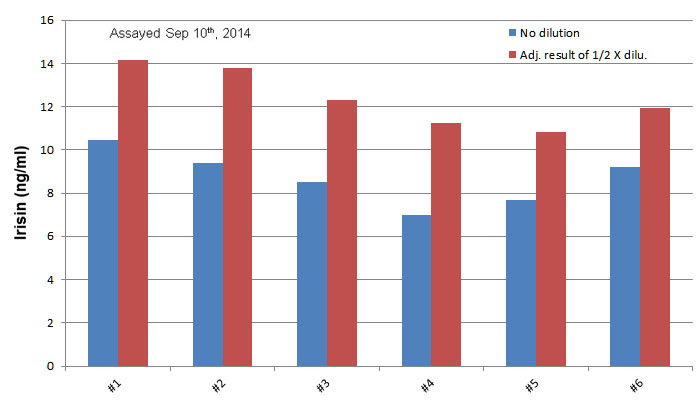
Irisin is induced in renal ischemia-reperfusion to protect against tubular cell injury via suppressing p53.
Liu Y, Fu Y, Liu Z, et al. Biochim Biophys Acta Mol Basis Dis. 2020;1866(7):165792.
Recombinant Irisin Prevents the Reduction of Osteoblast Differentiation Induced by Stimulated Microgravity through Increasing β-Catenin Expression.
Chen Z, Zhang Y, Zhao F, et al. Int J Mol Sci. 2020;21(4)
Irisin reverses intestinal epithelial barrier dysfunction during intestinal injury via binding to the integrin αVβ5 receptor.
Bi J, Zhang J, Ren Y, et al. J Cell Mol Med. 2020;24(1):996-1009.
Irisin promotes proliferation but inhibits differentiation in osteoclast precursor cells.
Ma Y, Qiao X, Zeng R, et al. FASEB J. 2018;:fj201700983RR.
Effects and underlying mechanisms of irisin on the proliferation and apoptosis of pancreatic β cells.
Liu S, Du F, Li X, et al. PLoS ONE. 2017;12(4):e0175498.
Exercise-indusced irisin in bone and systemic irisin administration reveal new regulatory mechanisms of bone metabolism.
Zhang J, Valverde P, Zhu X, et al. Bone Research. 2017;5:16056.
Irisin Lowers Blood Pressure by Improvement of Endothelial Dysfunction via AMPK-Akt-eNOS-NO Pathway in the Spontaneously Hypertensive Rat.
Fu J, Han Y, Wang J, et al. J Am Heart Assoc. 2016;5(11)
FNDC5 expression and circulating irisin levels are modified by diet and hormonal conditions in hypothalamus, adipose tissue and muscle.
Varela-rodríguez BM, Pena-bello L, Juiz-valiña P, Vidal-bretal B, Cordido F, Sangiao-alvarellos S. Sci Rep. 2016;6:29898.
Irisin evokes bradycardia by activating cardiac-projecting neurons of nucleus ambiguus.
Brailoiu E, Deliu E, Sporici RA, Brailoiu GC. Physiol Rep. 2015;3(6)
Central and peripheral irisin differentially regulate blood pressure.
Zhang W, Chang L, Zhang C, et al. Cardiovasc Drugs Ther. 2015;29(2):121-7.
Irisin, a Novel Myokine, Regulates Glucose Uptake in Skeletal Muscle Cells via AMPK.
Lee HJ, Lee JO, Kim N, et al. Mol Endocrinol. 2015;29(6):873-81.
Social Network Confirmation