OBJECTIVE: Pulmonary fibrosis causes high morbidity and mortality in affected individuals. Recently, we showed that parenteral or intratracheal administration of a peptide derived from endostatin, called E4, prevents and ameliorates fibrosis using different models of dermal and pulmonary disease. No marketed orally delivered peptide drugs are currently available for progressive pulmonary fibrosis; however oral delivery of drugs is the preferred route for treating most chronic diseases. Thus, we investigated whether oral administration of E4 peptide exerted anti-fibrotic activity in a murine pulmonary fibrosis model.
METHODS: Bleomycin (1.2mU/g body weight) was intratracheally administrated to male 6-8-week-old C57BL/6J mice. E4 peptide (20, 10, 5, and 1?g/mouse) or scrambled control peptide (20?g/mouse) was orally administered on the same day as bleomycin. In some experiments, E4 peptide (10 and 5?g/mouse) was orally administered three times on days 0, 3, and 6 post-bleomycin treatment. Lungs were harvested on day 21 for histological analysis and hydroxyproline assay.
RESULTS: Histological analysis and hydroxyproline assay revealed that bleomycin successfully induced pulmonary fibrosis, and that 20?g of oralE4 peptide ameliorated the fibrosis. The lower doses of E4 peptide (10, 5, and 1?g) were insufficient to exert anti-fibrotic activity when given as a single dose. Multiple doses of E4 peptide efficiently exerted anti-fibrotic activity even at lower doses.
CONCLUSION: E4 peptide shows oral bioavailability and exerts anti-fibrotic activity in a bleomycin-induced pulmonary fibrosis model. We suggest that E4 peptide is a novel oral drug for fibroproliferative disorders.
Nishimoto T, Mlakar L, Takihara T, Feghali-Bostwick C, Int Immunopharmacol. 2015 Oct;28(2):1102-5. doi: 10.1016/j.intimp.2015.07.039. Epub 2015 Aug 24.
Fibroproliferative disorders such as idiopathic pulmonary fibrosis and systemic sclerosis have no effective therapies and result in significant morbidity and mortality due to progressive organ fibrosis. We examined the effect of peptides derived from endostatin on existing fibrosis and fibrosis triggered by two potent mediators, transforming growth factor-? (TGF-?) and bleomycin, in human and mouse tissues in vitro, ex vivo, and in vivo. We identified one peptide, E4, with potent antifibrotic activity. E4 prevented TGF-?-induced dermal fibrosis in vivo in a mouse model, ex vivo in human skin, and in bleomycin-induced dermal and pulmonary fibrosis in vivo, demonstrating that E4 exerts potent antifibrotic effects. In addition, E4 significantly reduced existing fibrosis in these preclinical models. E4 amelioration of fibrosis was accompanied by reduced cell apoptosis and lower levels of lysyl oxidase, an enzyme that cross-links collagen, and Egr-1 (early growth response gene-1), a transcription factor that mediates the effects of several fibrotic triggers. Our findings identify E4 as a peptide with potent antifibrotic activity and a possible therapeutic agent for organ fibrosis.
Yamaguchi Y, Takihara T, Chambers RA, Sci Transl Med. 2012 May 30;4(136):136ra71. doi: 10.1126/scitranslmed.3003421.
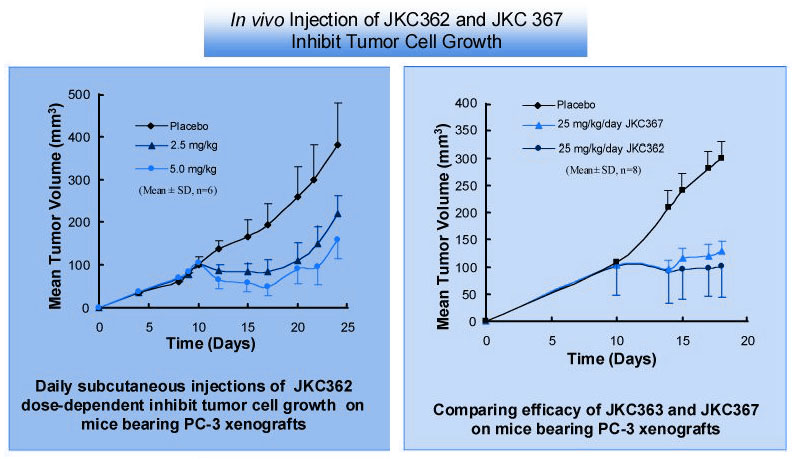
The angiogenesis inhibitor Endostatin is a 20 KDa carboxyl-terminal fragment of collagen XVIII. This compound has generated a great deal of excitement in the lay community as well as in the scientific community because of its potential as an anticancer therapy that, to date, will not induce acquired drug resistance. we report the synthesis and in vitro and in vivo testing of two water-soluble fragments of Endostatin with potent anti-tumor activity in vivo that have no effect on tumor cells in vitro. One peptide is a stable 63-residue polypeptides of 6985 M. W. called JKC-362 (human Endostatin (52-114)-NH2 ) that does not contain the required zinc-binding domain of Endostatin, but retains the ability to reduce established termed JKC-367, also inhibits the expansion of human tumor xenografts. JKC-362 and JKC-367, therefore, have similar anticancer therapeutic potential, but lack a critical domain for Endostatin's function. This is the first demonstration of a biological response to fragments of Endostatin, and may have important implications in the elucidation of the mechanism by which Endostatin inhibits angiogenesis.
J.D. Hunt, A. W. Zieske, N. Skrepnik et al., Proceedings of the American Association for Cancer Research, 41,487(3106), 2000
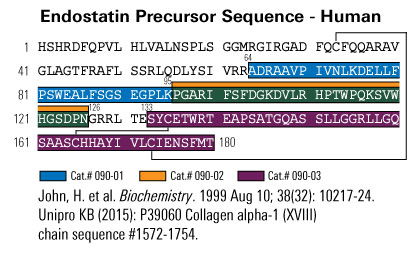
Circulating elongated forms of the angiogenesis inhibitor and potential anti-cancer drug endostatin were isolated from human blood filtrate. Immunoreactive endostatin was identified by a polyclonal rabbit antiserum raised against an N-terminal epitope of the polypeptide and purified by consecutive chromatographic steps and immunoblotting. N- and C-terminal sequence analyses of the isolated molecules revealed different forms of endostatin starting with V(117)HLRPAR. lacking the last and final three residues of the noncollagenous domain 1 (NC-1) of collagen XVIII, respectively. These polypetides are found to be O-glycosylated at T(125) (residue 9) with a glycan structure of the mucin type consisting of galactose N-acetylgalactosamine and N-acetylneuraminic acid residues. Carbohydrate analyses were performed via the semiquantitative HPLC-electrospray ionization mass spectrometry (ESMS) technique after exoglycosidase hydrolysis. Circulating endostatins are present as sialoglycoprotein (22,000 and 21,841Da +/- 0.02%) and asialoglycoprotein structures (21,710 and 21,549Da +/- 0.02%), while the two completely deglycosylated forms are obtained only after enzymatic incubation. The described glycosylated endostatins may represent intermediates in the proteolytic pathway of the NC-1 domain of collagen XVIII resulting in bioactive endostatins. Furthermore, immunoreactive endostatin-related C-terminal fragments of human collagen XV are found in the hemofiltrate. These polypeptides exhibit the N-terminal sequences P(66)HLLPPP. and Y(81)EKPALH. of the collagen XV NC-1 domain. ESMS and immunoblotting analyses reveal three glycosylated polypeptides with a molecular mass ranging from 16 to 21 kDa. Due to the high degree of homology between collagen XV and collagen XVIII as well as their analoqous proteolytic processing, functional similarities of collagen XVIII- and XV-related fragments should be revealed in future experiments.
John H, Preissner KT, Forssmann WG, Ständker L. Biochemistry. 1999 Aug 10;38(32):10217-24.
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 090-03 | Endostatin (133-180) amide / Endostatin Derived E4 (Human) | 100 µg | $444 |
| 090-01 | Endostatin (52-114) amide / JKC362 (Human) | 100 µg | $242 |
| G-090-01 | Endostatin (52-114) amide / JKC362 (Human) - Purified IgG Antibody | 200 µg | $571 |
| 090-02 | Endostatin (84-114) amide / JKC-367 (Human) | 100 µg | $167 |
Social Network Confirmation