

OBJECTIVE: To test the hypothesis that insulin-like growth factors-I and II (IGF-I and II) decline during late midlife and that greater declines are related to higher fat mass and lower lean mass.
METHODS: A total of 1,542 men and women in a British birth cohort study had IGF-I and II measured by immunoassay of blood samples at age 53 and/or 60-64 years. Fat mass, android: gynoid fat ratio, and appendicular lean mass were measured at 60-64 years using dual-energy X-ray absorptiometry (DXA). Associations between changes in IGF-I or II and body composition outcomes were examined using conditional change linear regression models.
RESULTS: Mean IGF-I and IGF-II concentrations were lower at 60-64 than at 53 years, by 12.8% for IGF-I and by 12.5% for IGF-II. Larger declines in either IGF-I or II were associated with higher fat mass at 60-64 years. Although higher IGF-I at 53 years was associated with higher lean mass, there was little evidence linking changes in IGF-I or II to lean mass. CONCLUSIONS: The findings suggest that IGF-I and II concentrations decline with age, and greater declines are associated with higher fat mass levels. These results provide some evidence for the suggested roles of IGF-I and II in regulating fat mass but not lean mass in older age.
Bann D, Holly JM, Lashen H et al., Obesity (Silver Spring). 2015 Feb 3. doi: 10.1002/oby.21002. [Epub ahead of print]
The extensive intratumor heterogeneity revealed by sequencing cancer genomes is an essential determinant of tumor progression, diagnosis, and treatment. What maintains heterogeneity remains an open question because competition within a tumor leads to a strong selection for the fittest subclone. Cancer cells also cooperate by sharing molecules with paracrine effects, such as growth factors, and heterogeneity can be maintained if subclones depend on each other for survival. Without strict interdependence between subclones, however, nonproducer cells can free-ride on the growth factors produced by neighboring producer cells, a collective action problem known in game theory as the "tragedy of the commons," which has been observed in microbial cell populations. Here, we report that similar dynamics occur in cancer cell populations. Neuroendocrine pancreatic cancer (insulinoma) cells that do not produce insulin-like growth factor II (IGF-II) grow slowly in pure cultures but have a proliferation advantage in mixed cultures, where they can use the IGF-II provided by producer cells. We show that, as predicted by evolutionary game theory, producer cells do not go extinct because IGF-II acts as a nonlinear public good, creating negative frequency-dependent selection that leads to a stable coexistence of the two cell types. Intratumor cell heterogeneity can therefore be maintained even without strict interdependence between cell subclones. Reducing the amount of growth factors available within a tumor may lead to a reduction in growth followed by a new equilibrium, which may explain relapse in therapies that target growth factors. F-II production maintained by public goods dynamics in neuroendocrine pancreatic cancer.
Archetti M, Ferraro DA, Christofori G. Heterogeneity for IGF-II production maintained by public goods dynamics in neuroendocrine pancreatic cancer. Proc Natl Acad Sci USA. 2015;112(6):1833-8.
A 45-year-old man was referred to our hospital and found to have a tubular adenocarcinoma of the descending colon with multiple liver metastases. During hospitalization, the patient suffered recurrent hypoglycemic attacks that required intravenous 50 % glucose infusion. He was diagnosed with non-islet cell tumor hypoglycemia (NICTH) because the colon cancer tissue obtained by biopsy was strongly stained for insulin-like growth factor-II(IGF-II) by immunohistochemistry. He received chemotherapy with oxaliplatin, 5-FU and leucovorin (FOLFOX) plus bevacizumab (Bmab), and showed a partial response. As the metastatic lesions decreased in size, the hypoglycemic attacks gradually disappeared. Subsequently, he received outpatient chemotherapy and maintained a high quality of life for about 10 months. Western blot analysis of IGF-II in serum at the time of admission showed a high-molecular-weight form of IGF-II, which was considered to have caused hypoglycemia. This patient presents a very rare case of colorectal cancer associated with NICTH syndrome due to production of high-molecular-weight IGF-II by cancer cells. It is important to investigate IGF-II expression in cancer tissues for establishing the diagnosis of NICTH in cases with intractable hypoglycemia complicated by advanced cancer.
Teramae S, Miyamoto H, Muguruma N, et al. Insulin-like growth factor II-producing metastatic colon cancer with recurrent hypoglycemia. Clin J Gastroenterol. 2015;8(1):35-40.
IGF2 is an autocrine ligand for the beta cell IGF1R receptor and GLP-1 increases the activity of this autocrine loop by enhancing IGF1R expression, a mechanism that mediates the trophic effects of GLP-1 on beta cell mass and function. Here, we investigated the regulation of IGF2 biosynthesis and secretion. We showed that glutamine rapidly and strongly induced IGF2 mRNA translation using reporter constructs transduced in MIN6 cells and primary islet cells. This was followed by rapid secretion of IGF2 via the regulated pathway, as revealed by the presence of mature IGF2 in insulin granule fractions and by inhibition of secretion by nimodipine and diazoxide. When maximally stimulated by glutamine, the amount of secreted IGF2 rapidly exceeded its initial intracellular pool and tolbutamide, and high K(+) increased IGF2 secretion only marginally. This indicates that the intracellular pool of IGF2 is small and that sustained secretion requires de novo synthesis. The stimulatory effect of glutamine necessitates its metabolism but not mTOR activation. Finally, exposure of insulinomas or beta cells to glutamine induced Akt phosphorylation, an effect that was dependent on IGF2 secretion, and reduced cytokine-induced apoptosis. Thus, glutamine controls the activity of the beta cell IGF2/IGF1R autocrine loop by increasing the biosynthesis and secretion of IGF2. This autocrine loop can thus integrate changes in feeding and metabolic state to adapt beta cell mass and function.
Modi H, Cornu M, Thorens B. Glutamine stimulates biosynthesis and secretion of insulin-like growth factor 2 (IGF2), an autocrine regulator of beta cell mass and function. J Biol Chem. 2014;289(46):31972-82.
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 033-23 | IGF-II, recombinant (Human) | 20 µg | $133 |
| T-033-23 | IGF-II, recombinant (Human) - I-125 Labeled | 10 µCi | $1322 |
| 035-19 | pro-Insulin-Like Growth Factor 2 (IGF-2) (105-128) (Human) | 200 µg | $69 |
| 035-20 | pro-Insulin-Like Growth Factor 2 (IGF-2) (131-154) (Human) | 200 µg | $69 |
| 035-14 | Insulin-Like Growth Factor 2 (IGF-2) (33-40) (Human) | 1 mg | $108 |
| 035-16 | Insulin-Like Growth Factor 2 (IGF-2) (54-67) (Human) | 1 mg | $108 |
| 035-18 | pro-Insulin-Like Growth Factor 2 (IGF-2) (68-102) (Human) | 200 µg | $133 |
| 035-17 | Insulin-Like Growth Factor 2 (IGF-2) (69-84) (Human) | 200 µg | $128 |
| 035-13 | Insulin-Like Growth Factor 2 (IGF-2), D-Domain (62-67) (Human) | 1 mg | $108 |
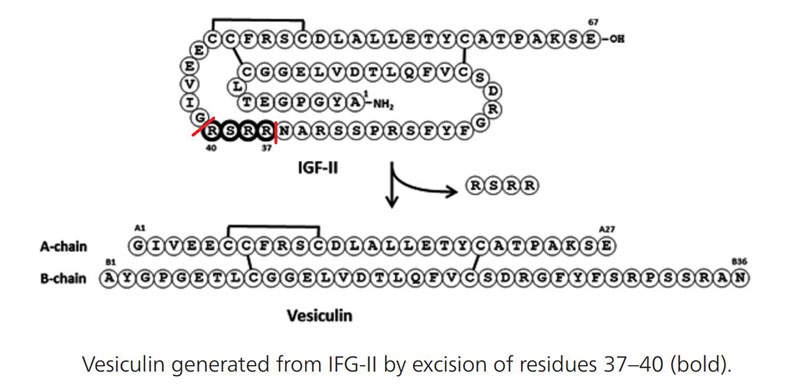
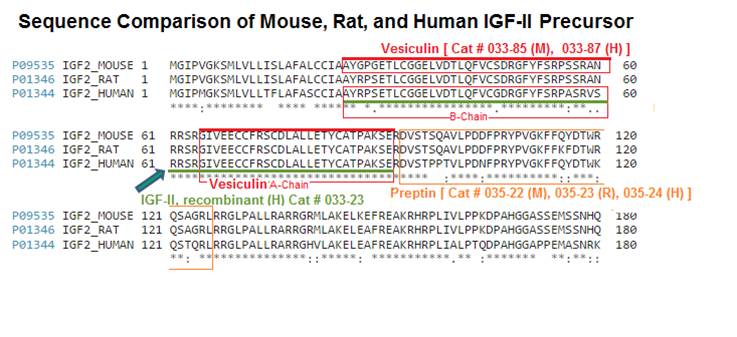
| 033-87 | Vesiculin / Des (37-40) IGF-II (Human) | 100 µg | $444 |
Social Network Confirmation