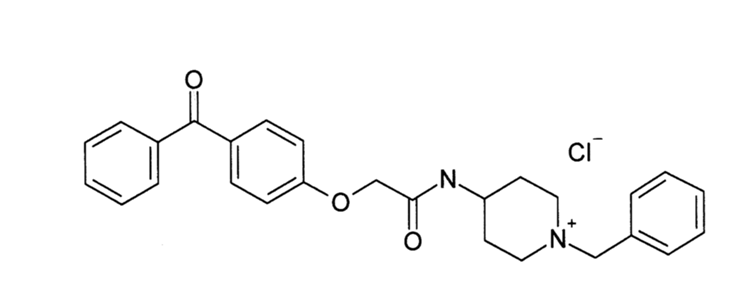
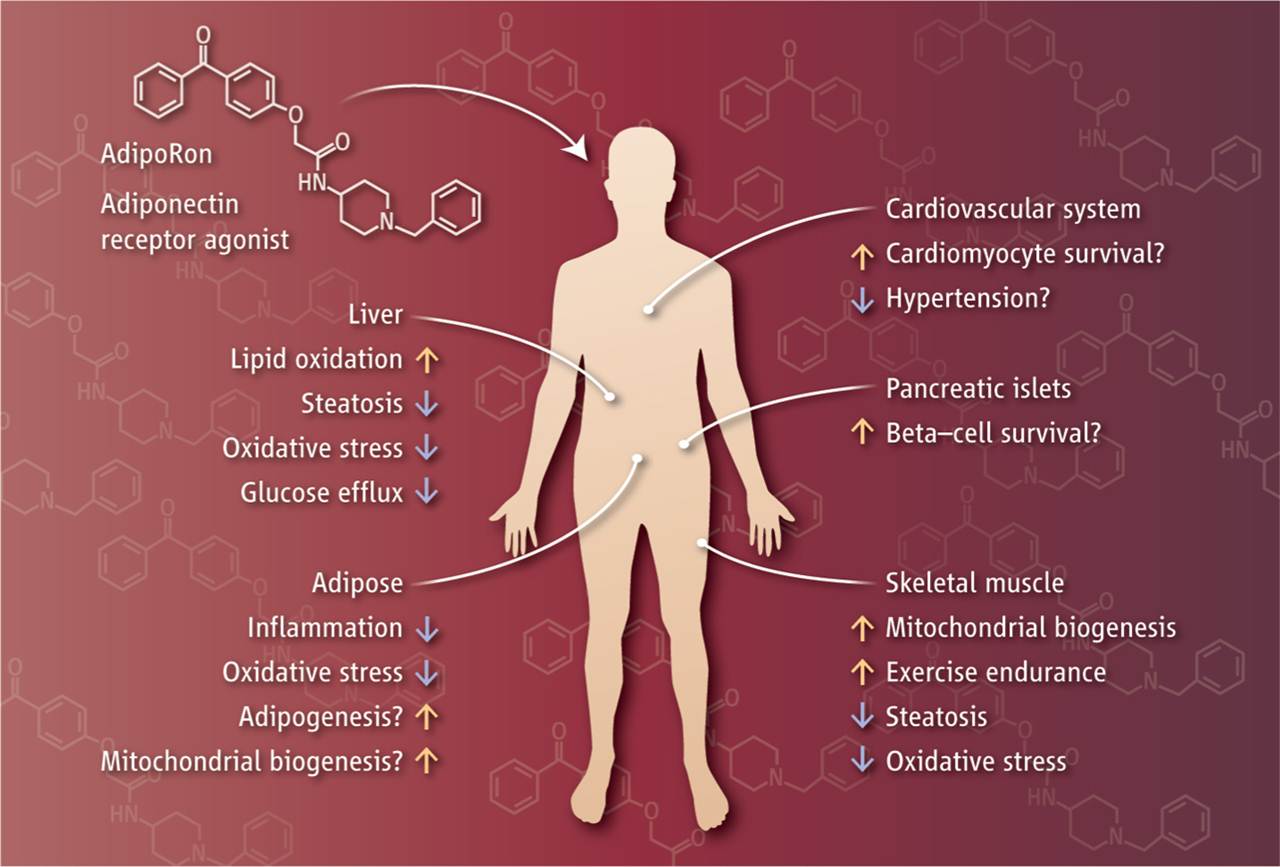
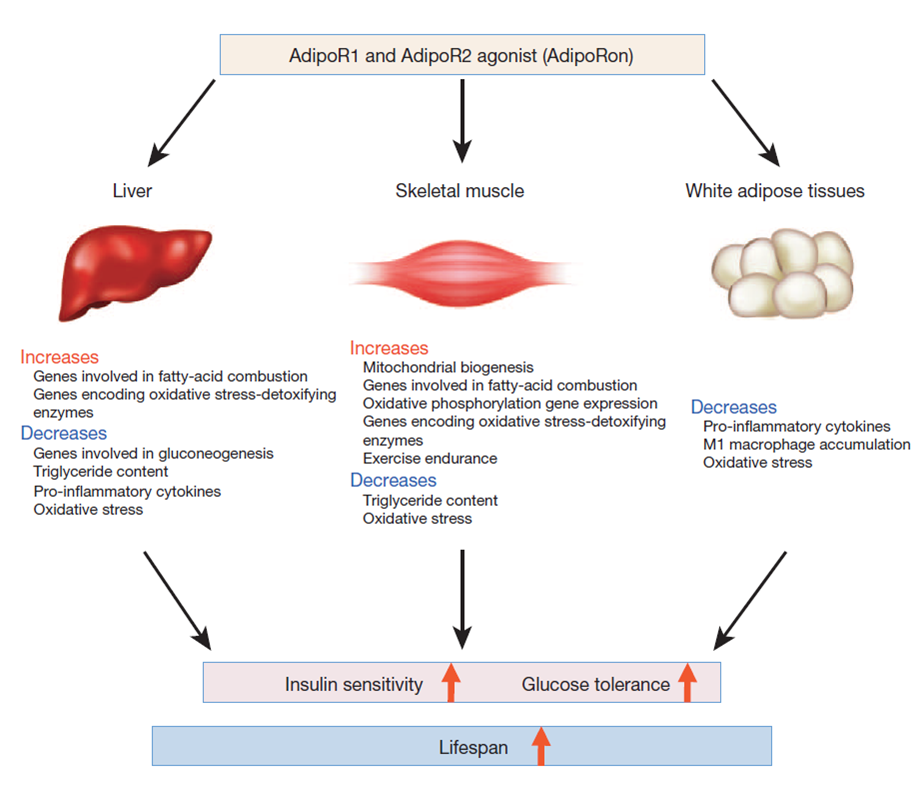
Adiponectin secreted from adipocytes binds to adiponectin receptors AdipoR1 and AdipoR2, and exerts antidiabetic effects via activation of AMPK and PPAR-? pathways, respectively. Levels of adiponectin in plasma are reduced in obesity, which causes insulin resistance and type 2 diabetes. Thus, orally active small molecules that bind to and activate AdipoR1 and AdipoR2 could ameliorate obesity-related diseases such as type 2 diabetes. Here we report the identification of orally active synthetic small-molecule AdipoR agonists. One of these compounds, AdipoR agonist (AdipoRon), bound to both AdipoR1 and AdipoR2 in vitro. AdipoRon showed very similar effects to adiponectin in muscle and liver, such as activation of AMPK and PPAR-? pathways, and ameliorated insulin resistance and glucose intolerance in mice fed a high-fat diet, which was completely obliterated in AdipoR1 and AdipoR2 double-knockout mice. Moreover, AdipoRon ameliorated diabetes of genetically obese rodent model db/db mice, and prolonged the shortened lifespan of db/db mice on a high-fat diet. Thus, orally active AdipoR agonists such as AdipoRon are a promising therapeutic approach for the treatment of obesity-related diseases such as type 2 diabetes.
Okada-iwabu M, Yamauchi T, Iwabu M, et al. A small-molecule AdipoR agonist for type 2 diabetes and short life in obesity. Nature. 2013;503(7477):493-9.
Almost 20 years have passed since the first laboratory evidence emerged that an abundant message encoding a protein with homology to the C1q superfamily is highly specifically expressed in adipocytes. At this stage, we refer to this protein as adiponectin. Despite more than 10,000 reports in the literature since its initial description, we seem to have written only the first chapter in the textbook on adiponectin physiology. With every new aspect we learn about adiponectin, a host of new questions arise with respect to the underlying molecular mechanisms. Here, we aim to summarize recent findings in the field and bring the rodent studies that suggest a causal relationship between adiponectin levels in plasma and systemic insulin sensitivity in perspective with the currently available data on the clinical side.
Risheng Ye, Philipp E. Scherer, Molecular Metabolism, Volume 2, Issue 3, August 2013, Pages 133-141
The great interest that scientists have for adiponectin is primarily due to its central metabolic role. Indeed, the major function of this adipokine is the control of glucose homeostasis that it exerts regulating liver and muscle metabolism. Adiponectin has insulin-sensitizing action and leads to down-regulation of hepatic gluconeogenesis and an increase of fatty acid oxidation. In addition, adiponectin is reported to play an important role in the inhibition of inflammation. The hormone is secreted in full-length form, which can either assemble into complexes or be converted into globular form by proteolytic cleavage. Over the past few years, emerging publications reveal a more varied and pleiotropic action of this hormone. Many studies emphasize a key role of adiponectin during tissue regeneration and show that adiponectin deficiency greatly inhibits the mechanisms underlying tissue renewal. This review deals with the role of adiponectin in tissue regeneration, mainly referring to skeletal muscle regeneration, a process in which adiponectin is deeply involved. In this tissue, globular adiponectin increases proliferation, migration and myogenic properties of both resident stem cells (namely satellite cells) and non-resident muscle precursors (namely mesoangioblasts). Furthermore, skeletal muscle could be a site for the local production of the globular form that occurs in an inflamed environment. Overall, these recent findings contribute to highlight an intriguing function of adiponectin in addition to its well-recognized metabolic action.
Fiaschi T, Magherini F, Gamberi T, Modesti PA, Modesti A. Adiponectin as a tissue regenerating hormone: more than a metabolic function. Cell Mol Life Sci. 2014;71(10):1917-25.
Adipokines, that is factors secreted by adipose tissue, act through a network of autocrine, paracrine, and endocrine pathways to regulate several aspects of physiology, including glucose and lipid metabolism, neuroendocrine function, reproduction, and cardiovascular function. In particular, adiponectin, a 30-kDa protein, is associated with the regulation of insulin sensitivity, and its levels in serum are affected by altered metabolic homeostasis. Adiponectin effects are mediated by adiponectin receptors, which occur as two isoforms (AdipoR1 and AdipoR2). Transcriptional regulation of adiponectin is by the peroxisome proliferator-activated receptor-gamma (PPAR-γ). However, acting through AdipoR1 and AdipoR2, adiponectin enhances 5' adenosine monophosphate-activated protein kinase (AMPK) and the PPARα-mediated pathways in the liver and skeletal muscles. Adiponectin receptors mediate a wide spectrum of metabolic reactions, including gluconeogenesis and fatty-acid oxidation. Altogether, adiponectin deficiency and/or decreased adiponectin receptor-mediated activity possibly contribute to insulin resistance in metabolic syndromes, coronary heart disease, and liver disease.
Akingbemi BT. Adiponectin receptors in energy homeostasis and obesity pathogenesis. Prog Mol Biol Transl Sci. 2013;114:317-42.
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 001-16 | AdipoRon / Adiponectin Receptor Antagonist | 1 mg | $102 |
Social Network Confirmation