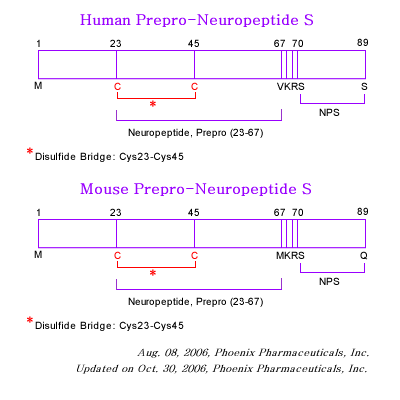
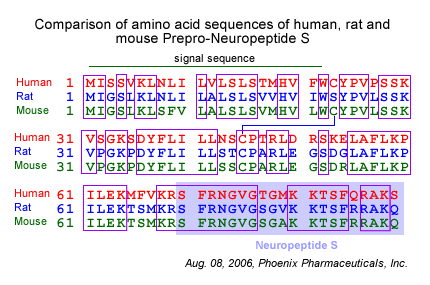


Neuropeptide S (NPS) and its receptor (NPSR) are thought to have a role in asthma pathogenesis; a number of single nucleotide polymorphisms within NPSR have been shown to be associated with an increased prevalance of asthma. One such single nucleotide polymorphism leads to the missense mutation N107I, which results in an increase in the potency of NPS for NPSR. To gain insight into structure-function relationships within NPS and NPSR, we first carried out a limited structural characterization of NPS and subjected the peptide to extensive mutagenesis studies. Our results show that the NH(2)-terminal third of NPS, in particular residues Phe-2, Arg-3, Asn-4, and Val-6, are necessary and sufficient for activation of NPSR. Furthermore, part of a nascent helix within the peptide, spanning residues 5 through 13, acts as a regulatory region that inhibits receptor activation. Notably, this inhibition is absent in the asthma-linked N107I variant of NPSR, suggesting that residue 107 interacts with the aforementioned regulatory region of NPS. Whereas this interaction may be at the root of the increase in potency associated with the N107I variant, we show here that the mutation also causes an increase in cell-surface expression of the mutant receptor, leading to a concomitant increase in the maximal efficacy (E(max)) of NPS. Our results identify the key residues of NPS involved in NPSR activation and suggest a molecular basis for the functional effects of the N107I mutation and for its putative pathophysiological link with asthma.
Bernier V, Stocco R, Bogusky MJ, et al. Structure-function relationships in the neuropeptide S receptor: molecular consequences of the asthma-associated mutation N107I. J Biol Chem. 2006;281(34):24704-12.
Many different neuropharmacological agents modulate arousal and anxiety, yet to date, few endogenous substances have produced arousal with an anxiolytic effect. In this issue of Neuron, Xu et al. describe the localization and characterization of a novel neuropeptide, neuropeptide S (and its cognate receptor), that is unique in its arousing and anxiolytic-like properties.
Koob GF, Greenwell TN. Neuropeptide S: a novel activating anxiolytic?. Neuron. 2004;43(4):441-2.
Arousal and anxiety are behavioral responses that involve complex neurocircuitries and multiple neurochemical components. Here, we report that a neuropeptide, neuropeptide S (NPS), potently modulates wakefulness and could also regulate anxiety. NPS acts by activating its cognate receptor (NPSR) and inducing mobilization of intracellular Ca2+. The NPSR mRNA is widely distributed in the brain, including the amygdala and the midline thalamic nuclei. Central administration of NPS increases locomotor activity in mice and decreases paradoxical (REM) sleep and slow wave sleep in rats. NPS was further shown to produce anxiolytic-like effects in mice exposed to four different stressful paradigms. Interestingly, NPS is expressed in a previously undefined cluster of cells located between the locus coeruleus (LC) and Barrington's nucleus. These results indicate that NPS could be a new modulator of arousal and anxiety. They also show that the LC region encompasses distinct nuclei expressing different arousal-promoting neurotransmitters.
Xu YL, Reinscheid RK, Huitron-resendiz S, et al. Neuropeptide S: a neuropeptide promoting arousal and anxiolytic-like effects. Neuron. 2004;43(4):487-97.
Pharmacological characteristics of NPS Receptor and Neuropeptides |
||||
| Binding of NPS Receptor | ||||
| Number of Samples | FLIPR (EC50, nM) | (IC50, nM) | kd (nM) | Bmax (fmol/150000 cells) |
| Human NPS | 9.4 ± 3.2 | 0.42 ± 0.12 | ||
| Rat NPS | 3.2 ± 1.1 | |||
| Mouse NPS | 3.0 ± 1.3 | |||
| Human NPS, 125I-Tyr10 | 6.7 ± 2.4 | 0.33 ± 0.12 | 3.2 ± 0.4 | |
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| B-005-88 | Neuropeptide S (Rat) - Biotin Labeled | 10 µg | $382 |
| FC3-005-88 | Neuropeptide S (Rat) - Cy3 Labeled | 1 nmol | $714 |
| FG-005-88A | Neuropeptide S (Rat) - FAM Labeled | 1 nmol | $382 |
| T-005-86 | [Tyr15]-Neuropeptide S (Rat) - I-125 Labeled | 10 µCi | $1082 |
| FR-005-88 | Neuropeptide S (Rat) - Rhodamine Labeled | 1 nmol | $444 |
| 005-87 | [Tyr10]-Neuropeptide S (Rat, Mouse) | 100 µg | $167 |
| T-005-87 | [Tyr10]-Neuropeptide S (Rat, Mouse) - I-125 Labeled | 10 µCi | $1082 |
| 048-36 | Neuropeptide SF (NPSF) (1-35) (Bovine) | 100 µg | $189 |
| 048-32 | Neuropeptide SF (NPSF) (15-37) (Rat) | 200 µg | $242 |
| 048-34 | [Tyr30]-Neuropeptide SF (NPSF) (30-37) (Human) | 200 µg | $133 |
Social Network Confirmation