Bioactive Nesfatin-1-like peptide (NLP) for Obesity and Diabetes related study
Abstract
Prognostic Impact of Modulators of G proteins in Circulating Tumor Cells from Patients with Metastatic Colorectal Cancer.
The consequence of a loss of balance between G-protein activation and deactivation in cancers has been interrogated by studying infrequently occurring mutants of trimeric G-protein ?-subunits and GPCRs. Prior studies on members of a newly identified family of non-receptor guanine nucleotide exchange factors (GEFs), GIV/Girdin, Daple, NUCB1 and NUCB2 have revealed that GPCR-independent hyperactivation of trimeric G proteins can fuel metastatic progression in a variety of cancers. Here we report that elevated expression of each GEF in circulating tumor cells (CTCs) isolated from the peripheral circulation of patients with metastatic colorectal cancer is associated with a shorter progression-free survival (PFS). The GEFs were stronger prognostic markers than two other markers of cancer progression, S100A4 and MACC1, and clustering of all GEFs together improved the prognostic accuracy of the individual family members; PFS was significantly lower in the high-GEFs versus the low-GEFs groups [H.R = 5, 20 (95% CI; 2,15-12,57)]. Because nucleotide exchange is the rate-limiting step in cyclical activation of G-proteins, the poor prognosis conferred by these GEFs in CTCs implies that hyperactivation of G-protein signaling by these GEFs is an important event during metastatic progression, and may be more frequently encountered than mutations in G-proteins and/or GPCRs.
Barbazan J, Dunkel Y, Li H, et al. Prognostic Impact of Modulators of G proteins in Circulating Tumor Cells from Patients with Metastatic Colorectal Cancer. Sci Rep. 2016;6:22112.
Nesfatin-1-like peptide is a novel metabolic factor that suppresses feeding, and regulates whole-body energy homeostasis in male Wistar rats.
Nucleobindin-1 has high sequence similarity to nucleobindin-2, which encodes the anorectic and metabolic peptide, nesfatin-1. We previously reported a nesfatin-1-like peptide (NLP), anorectic in fish and insulinotropic in mice islet beta-like cells. The main objective of this research was to determine whether NLP is a metabolic regulator in male Wistar rats. A single intraperitoneal (IP) injection of NLP (100 ?g/kg BW) decreased food intake and increased ambulatory movement, without causing any change in total activity or energy expenditure when compared to saline-treated rats. Continuous subcutaneous infusion of NLP (100 ?g/kg BW) using osmotic mini-pumps for 7 days caused a reduction in food intake on days 3 and 4. Similarly, water intake was also reduced for two days (days 3 and 4) with the effect being observed during the dark phase. This was accompanied by an increased RER and energy expenditure. However, decreased whole-body fat oxidation, and total activity were observed during the long-term treatment (7 days). Body weight gain was not significantly different between control and NLP infused rats. The expression of mRNAs encoding adiponectin, resistin, ghrelin, cholecystokinin and uncoupling protein 1 (UCP1) were significantly upregulated, while leptin and peptide YY mRNA expression was downregulated in NLP-treated rats. These findings indicate that administration of NLP at 100 ?g/kg BW reduces food intake and modulates whole body energy balance. In summary, NLP is a novel metabolic peptide in rats.
Gawli K, Ramesh N, Unniappan S. PLoS ONE. 2017;12(5):e0178329.
Nucleobindin 1 binds to multiple types of pre-fibrillar amyloid and inhibits fibrillization.
During amyloid fibril formation, amyloidogenic polypeptides misfold and self assemble into soluble pre-fibrillar aggregates, i.e., protofibrils, which elongate and mature into insoluble fibrillar aggregates. An emerging class of chaperones, chaperone-like amyloid binding proteins (CLABPs), has been shown to interfere with aggregation of particular misfolded amyloid peptides or proteins. We have discovered that the calcium-binding protein nuclebindin-1 (NUCB1) is a novel CLABP. We show that NUCB1 inhibits aggregation of islet-amyloid polypeptide associated with type 2 diabetes mellitus, a-synuclein associated with Parkinson’s disease, transthyretin V30M mutant associated with familial amyloid polyneuropathy, and A?42 associated with Alzheimer’s disease by stabilizing their respective protofibril intermediates. Kinetic studies employing the modeling software AmyloFit show that NUCB1 affects both primary nucleation and secondary nucleation. We hypothesize that NUCB1 binds to the common cross-?-sheet structure of protofibril aggregates to “cap” and stabilize soluble macromolecular complexes. Transmission electron microscopy and atomic force microscopy were employed to characterize the size, shape and volume distribution of multiple sources of NUCB1-capped protofibrils. Interestingly, NUCB1 prevents Aβ42 protofibril toxicity in a cellular assay. NUCB1-stabilized amyloid protofibrils could be used as immunogens to prepare conformation-specific antibodies and as novel tools to develop screens for anti-protofibril diagnostics and therapeutics.
Bonito-oliva A, Barbash S, Sakmar TP, Graham WV. Sci Rep. 2017;7:42880.
Nesfatin-1-Like Peptide Encoded in Nucleobindin-1 in Goldfish is a Novel Anorexigen Modulated by Sex Steroids, Macronutrients and Daily Rhythm.
Nesfatin-1 is an 82 amino acid anorexigen encoded in a secreted precursor nucleobindin-2 (NUCB2). NUCB2 was named so due to its high sequence similarity with nucleobindin-1 (NUCB1). It was recently reported that NUCB1 encodes an insulinotropic nesfatin-1-like peptide (NLP) in mice. Here, we aimed to characterize NLP in fish. RT- qPCR showed NUCB1 expression in both central and peripheral tissues. Western blot analysis and/or fluorescence immunohistochemistry determined NUCB1/NLP in the brain, pituitary, testis, ovary and gut of goldfish. NUCB1 mRNA expression in goldfish pituitary and gut displayed a daily rhythmic pattern of expression. Pituitary NUCB1 mRNA expression was downregulated by estradiol, while testosterone upregulated its expression in female goldfish brain. High carbohydrate and fat suppressed NUCB1 mRNA expression in the brain and gut. Intraperitoneal injection of synthetic rat NLP and goldfish NLP at 10 and 100?ng/g body weight doses caused potent inhibition of food intake in goldfish. NLP injection also downregulated the expression of mRNAs encoding orexigens, preproghrelin and orexin-A, and upregulated anorexigen cocaine and amphetamine regulated transcript mRNA in goldfish brain. Collectively, these results provide the first set of results supporting the anorectic action of NLP, and the regulation of tissue specific expression of goldfish NUCB1.
Sundarrajan L, Blanco AM, Bertucci JI, Ramesh N, Canosa LF, Unniappan S. Sci Rep. 2016;6:28377.
Nucleobindin-1 encodes a nesfatin-1-like peptide that stimulates insulin secretion.
Nesfatin-1 (82 amino acid) is an anorexigenic and insulinotropic peptide encoded in a secreted precursor, nucleobindin-2 (NUCB2). Nucleobindin-1 (NUCB1) is a protein with very high sequence similarity to NUCB2. We hypothesized that a nesfatin-1 like peptide (NLP) is encoded in NUCB1, and this peptide is biologically active. In silico analysis found a signal peptide cleavage site at position 25 (Arginine) and 26 (Valine) preceding the NLP region in NUCB1 sequence, and potential proprotein convertase cleavage sites at Lys-Arg (KR), forming a 77 amino acid NLP. RT-PCR studies found NUCB1 mRNA in both pancreas and MIN6 cells. NUCB1-like immunoreactivity was detected in mouse insulinoma (MIN6) cells, and pancreatic islet beta cells of mice. In order to determine the biological activity of NLP, MIN6 cells were incubated with synthetic rat NLP. NLP (10nM and 100nM) upregulated preproinsulin mRNA expression and insulin secretion at 1h post-incubation. In identical experiments using MIN6 cells, a scrambled peptide based on the NLP sequence did not elicit any effects on preproinsulin mRNA expression or insulin secretion. From this result, it is clear that an intact NLP sequence is required for its biological activity. NLP appears as another endogenous insulinotropic peptide encoded in NUCB1.
Ramesh N, Mohan H, Unniappan S. Gen Comp Endocrinol. 2015 Apr 20. pii: S0016-6480(15)00111-2. doi: 10.1016/j.ygcen.2015.04.011. [Epub ahead of print]
Gonzalez R1, Mohan H, Unniappan S, Gen Comp Endocrinol. 2012 May 1;176(3):341-6. doi: 10.1016/j.ygcen.2011.11.021. Epub 2011 Dec 3.
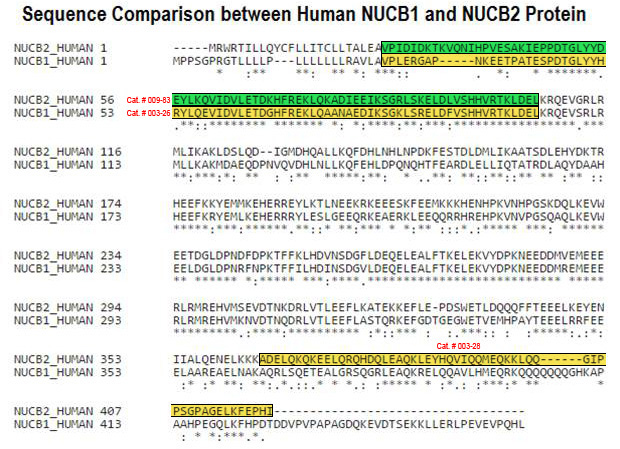
Schematics

More Information