PROTAC peptides open new possibilities for discovery of future therapies
Abstract
A PROTAC Peptide Induces Durable β-catenin Degradation and Suppresses Wnt-dependent Intestinal Cancer
Liao H, Li X, Zhao L, et al. A PROTAC peptide induces durable β-catenin degradation and suppresses Wnt-dependent intestinal cancer. Cell Discov. 2020;6(1):35.
A cell-permeable peptide-based PROTAC against the oncoprotein CREPT proficiently inhibits pancreatic cancer
Ma D, Zou Y, Chu Y, et al. A cell-permeable peptide-based PROTAC against the oncoprotein CREPT proficiently inhibits pancreatic cancer. Theranostics. 2020;10(8):3708-3721.
Assays and technologies for developing proteolysis targeting chimera degraders
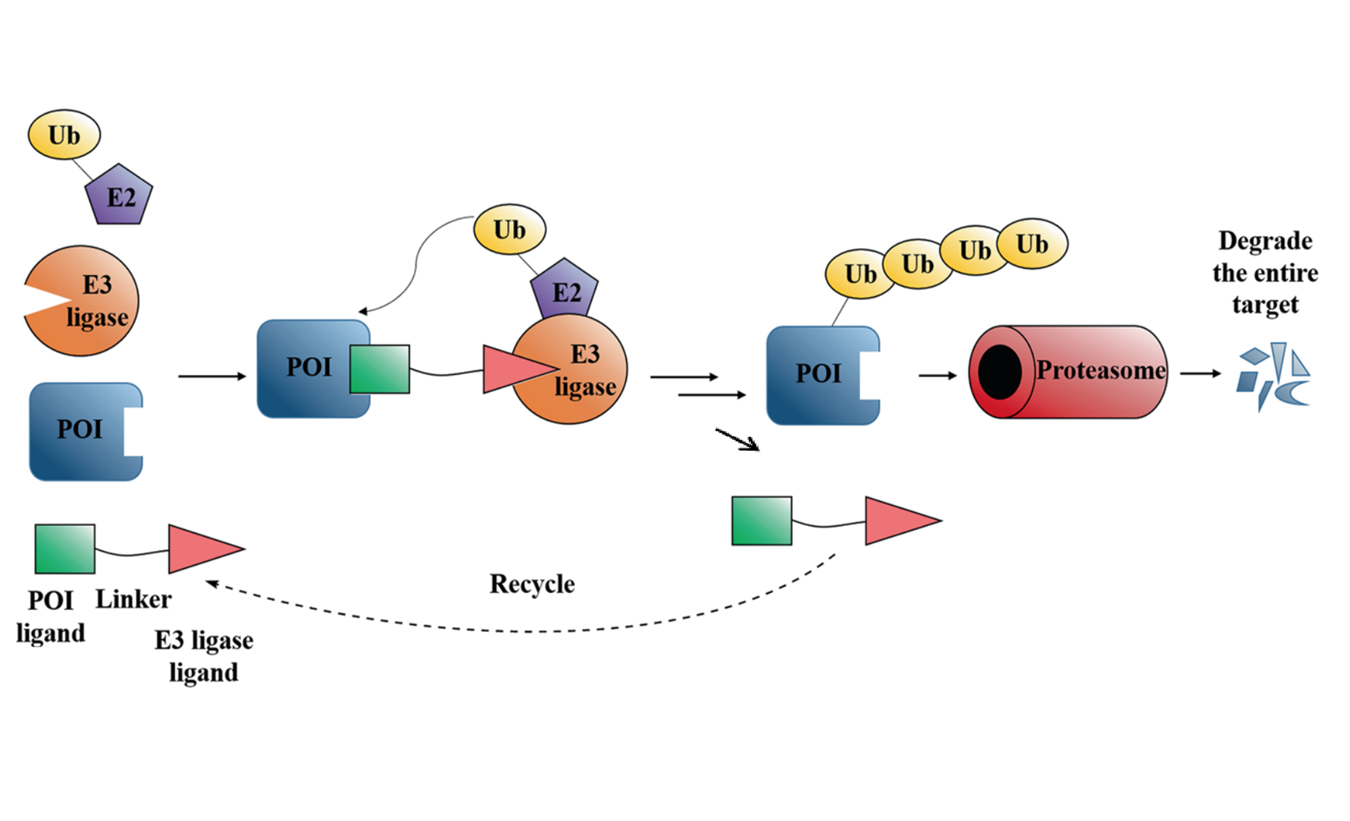
Abstract Targeted protein degradation by small-molecule degraders represents an emerging mode of action in drug discovery. Proteolysis targeting chimeras (PROTACs) are small molecules that can recruit an E3 ligase and a protein of interest (POI) into proximity, leading to induced ubiquitination and degradation of the POI by the proteasome system. To date, the design and optimization of PROTACs remain empirical due to the complicated mechanism of induced protein degradation. Nevertheless, it is increasingly appreciated that profiling step-by-step along the ubiquitin-proteasome degradation pathway using biochemical and biophysical assays are essential in understanding the structure-activity relationship and facilitating the rational design of PROTACs. This review aims to summarize these assays and to discuss the potential of expanding the toolbox with other new techniques.
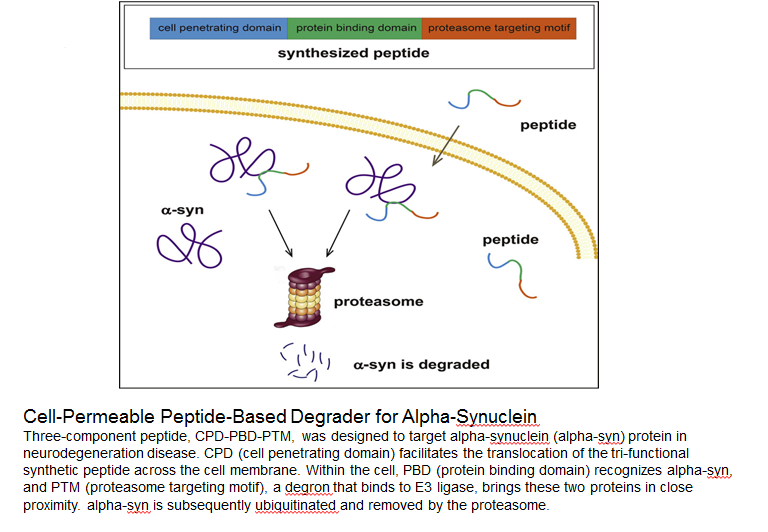
Specific Knockdown of α-Synuclein by Peptide-Directed Proteasome Degradation Rescued Its Associated Neurotoxicity
Qu J, Ren X, Xue F, et al. Specific knockdown of α-synuclein by peptide-directed proteasome degradation rescued its associated neurotoxicity. Cell Chemical Biology. 2020;27(6):751-762.e4.
PROTACs– a game-changing technology
PROTACs: great opportunities for academia and industry
Sun X, Gao H, Yang Y, et al. PROTACs: great opportunities for academia and industry. Signal Transduct Target Ther. 2019;4:64.
Discovery of a Keap1-dependent peptide PROTAC to knockdown Tau by ubiquitination-proteasome degradation pathway
Abstract: Induced protein degradation by PROTACs has emerged as a promising strategy to target nonenzymatic proteins inside the cell. The aim of this study was to identify Keap1, a substrate adaptor protein for ubiquitin E3 ligase involved in oxidative stress regulation, as a novel candidate for PROTACs that can be applied in the degradation of the nonenzymatic protein Tau. A peptide PROTAC by recruiting Keap1-Cul3 ubiquitin E3 ligase was developed and applied in the degradation of intracellular Tau. Peptide 1 showed strong in vitro binding with Keap1 and Tau. With proper cell permeability, peptide 1 was found to colocalize with cellular Keap1 and resulted in the coimmunoprecipitation of Tau and Keap1. The results of flow cytometry and western blotting assays showed that peptide 1 can downregulate the intracellular Tau level in both time- and concentration-dependent manner. The application of Keap1 siRNA silencing and the proteasome inhibitor MG132 confirmed that peptide 1 could promote the Keap1-dependent poly-ubiquitination and proteasome-dependent degradation of Tau. The results suggested that using PROTACs to recruit Keap1 to induce the degradation of Tau may show promising character in the treatment of neurodegenerative disease. Besides, our research demonstrated that Keap1 should be a promising E3 ligase adaptor to be used in the design of novel PROTACs.
Lu M, Liu T, Jiao Q, et al. Discovery of a Keap1-dependent peptide PROTAC to knockdown Tau by ubiquitination-proteasome degradation pathway. European Journal of Medicinal Chemistry. 2018;146:251-259.
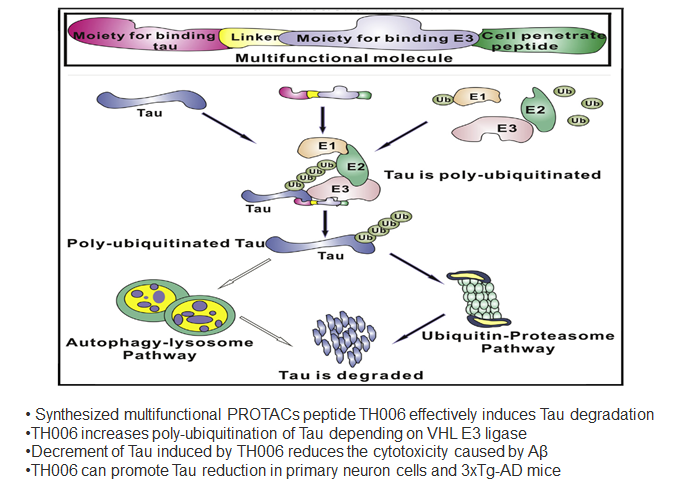
Specific Knockdown of Endogenous Tau Protein by Peptide-Directed Ubiquitin-Proteasome Degradation
Abstract: Tau, an important pathological protein of Alzheimer’s disease (AD), can mediate the toxicity of amyloid β (Aβ). Thus, reduction of Tau with chemical molecules may offer a novel strategy for treating AD. Here, we designed and synthesized a series of multifunctional molecules that contained Tau-recognition moieties and E3 ligase-binding moieties to enhance Tau degradation. Among these molecules, TH006 had the highest activity of inducing Tau degradation by increasing its poly-ubiquitination. The decrement in Tau induced by TH006 could decrease the cytotoxicity caused by Aβ. Furthermore, TH006 could regulate the Tau level in the brain of an AD mouse model. Therefore, partial reduction of Tau with such multifunctional peptides may open up a novel therapeutic strategy for AD treatment.
Chu T-T, Gao N, Li Q-Q, et al. Specific knockdown of endogenous tau protein by peptide-directed ubiquitin-proteasome degradation. Cell Chemical Biology. 2016;23(4):453-461.
Schematics