





Simultaneous expression of highly homologous RLN1 and RLN2 genes in prostate impairs their accurate delineation. We used PacBio SMRT sequencing and RNA-Seq in LNCaP cells in order to dissect the expression of RLN1 and RLN2 variants. We identified a novel fusion transcript comprising the RLN1 and RLN2 genes and found evidence of its expression in the normal and prostate cancer tissues. The RLN1-RLN2 fusion putatively encodes RLN2 isoform with the deleted secretory signal peptide. The identification of the fusion transcript provided information to determine unique RLN1-RLN2 fusion and RLN1 regions. The RLN1-RLN2 fusion was co-expressed with RLN1 in LNCaP cells, but the two gene products were inversely regulated by androgens. We showed that RLN1 is underrepresented in common PCa cell lines in comparison to normal and PCa tissue. The current study brings a highly relevant update to the relaxin field, and will encourage further studies of RLN1 and RLN2 in PCa and broader.Tevz G, Mcgrath S, Demeter R, et al. Identification of a novel fusion transcript between human relaxin-1 (RLN1) and human relaxin-2 (RLN2) in prostate cancer. Mol Cell Endocrinol. 2016;420:159-68.
Follicle-stimulating hormone (FSH) stimulates the proliferation of immature Sertoli cells through the activation of PI3K/AKT/mTORC1 and MEK/ERK1/2 pathways. Mature Sertoli cells stop proliferating and respond to FSH by stimulating cAMP production. To gain insight into possible mechanisms involved in this switch as well as the impact of paracrine factors that stimulate cell proliferation, we analyzed the effects of FSH and relaxin on intracellular signaling pathways involved with proliferation and differentiation in Sertoli cells from 15-day-old rats, which are close to the transition between the two stages. FSH stimulated 3H-thymidine incorporation and cyclin D1 expression, changes associated with proliferation. In contrast, FSH inhibited AKT and ERK1/2 phosphorylation, activated cAMP production and induced changes in several cell cycle genes that were compatible with differentiation. Relaxin also stimulated 3H-thymidine incorporation but increased phosphorylation of ERK1/2 and AKT. When both hormones were added simultaneously, relaxin attenuated FSH-mediated inhibition of ERK1/2 and AKT phosphorylation and FSH-mediated activation of cAMP production. FSH but not relaxin increased CREB phosphorylation, and relaxin but not FSH shifted NF-κB expression from the cytoplasm to the nucleus. Relaxin did not inhibit the effects of FSH on inhibin α and Bcl2 expression. We propose that at this time of Sertoli cell development, FSH starts to direct cells to differentiation through activation of cAMP/CREB and inhibition of ERK1/2 and AKT pathways. Relaxin counteracts FSH signaling through the inhibition of cAMP and activation of ERK1/2, AKT and NF-κB, but does not block the differentiation process triggered by FSH.
This publication used human relaxin-2 peptide for intracellular signaling pathways study.
Nascimento AR, Macheroni C, Lucas TF, Porto CS, Lazari MF. Reproduction. 2016;152(6):613-628.
AIMS: We evaluated relaxin's behaviour during a haemodialytic session and the effects of its intradialytic variability on blood pressure.METHODS: We enrolled 25 patients and evaluated relaxin's levels during a haemodialytic session. We also dosed interdialytic relaxin and enrolled 10 healthy subjects and 16 patients with III stage chronic renal failure as controls.RESULTS: Haemodialyzed patients have relaxin's baseline concentrations higher than healthy controls, but lower than chronic patients. During the treatment, relaxin is removed; it increases again throughout the interdialytic phase. Furthermore, relaxin's pre- haemodialytic concentration positively and significantly correlates with systolic, diastolic, and mean BP; such correlations disappear at the end of the treatment.CONCLUSION: Relaxin's removal during the treatment may intervene in the pathogenesis of intradialytic hypertension. Hence, relaxin could be not only a new biomarker but also an active player in the intradialytic variations of blood pressure.
this Publication used the human relaxin-2 RIA Kit (RK-035-62) from Phoenix Pharmaceuticals for relaxin-2 serum sample level measurement.
Cernaro V, Lacquaniti A, Giorgianni G, Bolignano D, Buemi M. Modifications in relaxin's serum levels during acetate-free biofiltration (AFB): only a new biomarker?. Ital J Anat Embryol. 2013;118(1 Suppl):98-9.
BACKGROUND/AIMS: Pregnancy is characterized by vasodilatation and increased glomerular filtration rate (GFR), despite overstimulation of the renin angiotensin system (RAS). The mesangial cells (MCs) influences GFR and when cultured from pregnant rats displays refractoriness to Ang II. We evaluated the role of relaxin (RLX) and its receptor (RXFP1), nitric oxide (NO) and the AT2 receptor in this response.METHODS: MCs cultured from kidneys of virgin (V) and pregnant (P) Wistar rats were treated with RLX or AT2 receptor blocker PD123319 or NO synthase inhibitor L-NAME. After 24 hr, intracellular calcium concentration ([Ca]i) was recorded before and after the addition of Ang II.RESULTS: MCs from V group expressed AT2, RLX and RXFP1, whose levels were increased in P cells. Ang II induced a 150% increase in [Ca] i in the V cells and 85% (p<0.05) in the P cells. V cells treated with RLX displayed a similar response to that observed in P cells, suggesting that RLX can modulate the reactivity of the MCs to Ang II. L-NAME and PD123319 did not interfere in this response.CONCLUSION: Results suggest that RLX is a mediator of the refractoriness of the MCs to Ang II during pregnancy.This Publication used a Relaxin-2 peptide (035-62) from Phoenix Pharmaceuticals.
Carvalho LN, Cristovam PC, Passos CS, Boim MA. Mesangial cells cultured from pregnant rats display reduced reactivity to angiotensin II: the role of relaxin, nitric oxide and AT2 receptor. Cell Physiol Biochem. 2012;30(6):1456-64.
Human relaxin-2 (hereafter simply defined as "relaxin") is a 6-kDa peptidic hormone best known for the physiological role played during pregnancy in the growth and differentiation of the reproductive tract and in the renal and systemic hemodynamic changes. This factor can also be involved in the pathophysiology of arterial hypertension and heart failure, in the molecular pathways of fibrosis and cancer, and in angiogenesis and bone remodeling. It belongs to the relaxin peptide family, whose members comprehensively exert numerous effects through interaction with different types of receptors, classified as relaxin family peptide (RXFP) receptors (RXFP1, RXFP2, RXFP3, RXFP4). Research looks toward the in-depth examination and complete understanding of relaxin in its various pleiotropic actions. The intent is to evaluate the likelihood of employing this substance for therapeutic purposes, for instance in diseases where a deficit could be part of the underlying pathophysiological mechanisms, also avoiding any adverse effect. Relaxin is already being considered as a promising drug, especially in acute heart failure. A careful study of the different RXFPs and their receptors and the comprehension of all biological activities of these hormones will probably provide new drugs with a potential wide range of therapeutic applications in the near future.
Cernaro V, Lacquaniti A, Lupica R, et al., Med Res Rev. 2014 Jan;34(1):77-105. doi: 10.1002/med.21277. Epub 2013 Feb 11.
AIMS: Serelaxin (Relaxin 2) is effective in relieving dyspnoea and improving multiple outcomes in acute heart failure (AHF). Many AHF patients have preserved ejection fraction (HFpEF). Given the lack of evidence-based therapies in this population, we evaluated the effects of serelaxin according to EF in RELAX-AHF trial.METHODS AND RESULTS: RELAX-AHF randomized 1161 AHF patients to 48-h serelaxin (30 μg/kg/day) or placebo within 16 h from presentation. We compared the effects of serelaxin on efficacy endpoints, safety endpoints, and biomarkers of organ damage between preserved (≥50%) and reduced (<50%, HFrEF) EF. HFpEF was present in 26% of patients. Serelaxin induced a similar dyspnoea relief in HFpEF vs. HFrEF patients by visual analogue scale-area under the curve (VAS-AUC) through Day 5 [mean change, 461 (-195, 1117) vs. 397 (10, 783) mm h, P = 0.87], but had possibly different effects on the proportion of patients with moderately or markedly dyspnoea improvement by Likert scale at 6, 12, and 24 h [odds ratio for favourable response, 1.70 (0.98, 2.95) vs. 0.85 (0.62, 1.15), interaction P = 0.030]. No differences were encountered in the effect of serelaxin on short- or long-term outcome between HFpEF and HFrEF patients including cardiovascular death or hospitalization for heart/renal failure through Day 60, cardiovascular death through Day 180, and all-cause death through Day 180. Similar safety and changes in biomarkers (high-sensitivity troponin T, cystatin-C, and alanine/aspartate aminotransferases) were found in both groups.CONCLUSIONS: In AHF patients with HFpEF compared with those with HFrEF, serelaxin was well tolerated and effective in relieving dyspnoea and had a similar effect on short- and long-term outcome, including survival improvement.Filippatos G, Teerlink JR, Farmakis D et al., Eur Heart J. 2013 Dec 6. [Epub ahead of print]
INTRODUCTION: Early allograft dysfunction following lung transplantation is mainly an ischemia/reperfusion (IR) injury. We showed that relaxin-2 (relaxin, Phoenix Pharmaceuticals) exerts a protective effect in lung IR, attributable to decreases in endothelin-1 (ET-1) production, leukocyte recruitment, and free radical generation. Here, we summarize our investigations into relaxin's signalling.MATERIALS AND METHODS: Isolated rat lungs were perfused with vehicle or 5 nM relaxin (n=6-10 each). Thereafter, experiments were conducted in the presence of relaxin plus vehicle, the protein kinase A inhibitors H-89 and KT-5720, the NO synthase (NOS) inhibitor L-NAME, the iNOS inhibitor 1400W, the nNOS inhibitor SMTC, the extracellular signal-regulated kinase-1/2 (ERK-1/2) inhibitor PD-98059, the phosphatidylinositol-3 kinase (PI3K) inhibitor wortmannin,the endothelin type-B (ETB) antagonist A-192621, or the glucocorticoid receptor (GR) antagonist RU-486. After 90 min ischemia and 90 min reperfusion we determined wet-to-dry (W/D) weight ratio, mean pulmonary arterial pressure (MPAP), vascular release of ET-1, neutrophil elastase (NE), myeloperoxidase (MPO), and malondialdehyde (MDA). Primary rat pulmonary vascular cells were similarly treated.RESULTS: IR lungs displayed significantly elevated W/D ratios, MPAP, as well as ET-1, NE, MDA, and MPO. In the presence of relaxin, all of these parameters were markedly improved. This protective effect was completely abolished by L-NAME, 1400W, PD-98059, and wortmannin whereas neither PKA and nNOS inhibition nor ETB and GR antagonism were effective. Analysis of NOS gene expression and activity revealed that the relaxin-induced early and moderate iNOS stimulation is ERK-1/2-dependent and counter-balanced by PI3K. Relaxin-PI3K-related phosphorylation of a forkhead transcription factor, FKHRL1, paralleled this regulation. In pulmonary endothelial and smooth muscle cells, FKHRL1 was essential to relaxin-PI3K signalling towards iNOS.CONCLUSION: In this short-time experimental setting, relaxin protects against IR-induced lung injury via early and moderate iNOS induction, dependent on balanced ERK-1/2 and PI3K-FKHRL1 stimulation. These findings render relaxin a candidate drug for lung preservation.Alexiou K, Wilbring M, Matschke K, Dschietzig T., PLoS One. 2013 Sep 30;8(9):e75592. doi: 10.1371/journal.pone.0075592.
AIMS: Human relaxin-2 influences renal and cardiovascular functions. We investigated its effects on experimental endothelial dysfunction.METHODS AND RESULTS: Acetylcholine-mediated vasodilation of rat aortic rings, impaired by 48 h tumour necrosis factor-α (TNF-α) treatment, was dose-dependently improved by relaxin co-incubation, an effect sensitive to phosphatidylinositol 3-kinase (PI3K) inhibition and the glucocorticoid receptor (GR) antagonist RU-486. TNF increased endothelial nitric oxide synthase (eNOS) phosphorylation at Thr495 and decreased total eNOS expression and both basal and stimulated eNOS activity. Relaxin co-incubation did not affect eNOS expression but improved its activity via PI3K-dependent Thr495 dephosphorylation and Ser1177 phosphorylation, and additional Ser633 phosphorylation. Via GR, relaxin attenuated the TNF-related stimulation of endothelin-1 expression, superoxide and nitrotyrosine formation, and arginase II expression. Relaxin restored, via GR-CCAAT/enhancer-binding protein-β (c/EBP-β)-mediated promoter stimulation, the compromised expression of superoxide dismutase-1 (SOD1). In rat aortic endothelial cells, relaxinactivated protein kinase B (Akt) and repressed TNF-induced nuclear factor-κB and activator protein-1. Finally, the relevance of the different findings to the model used was proved by pharmacological interventions.CONCLUSION: Relaxin improved endothelial dysfunction by promoting eNOS activity, suppressing endothelin-1 and arginase-II expression, and up-regulating SOD1 via GR, GR-c/EBP-β, and PI3K-Akt pathways. This corroborates the notion that it functions as an endogenous and potentially therapeutic vasoprotector.
The publication used the human relaxin 2 peptide from Phoenix Pharmaceuticals for investigation dose-dependent vasodilation-restoration by aortic ring vascular assays
Dschietzig T, Brecht A, Bartsch C, Baumann G, Stangl K, Alexiou K. Cardiovasc Res. 2012;95(1):97-107.
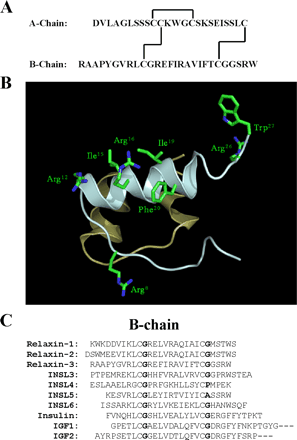
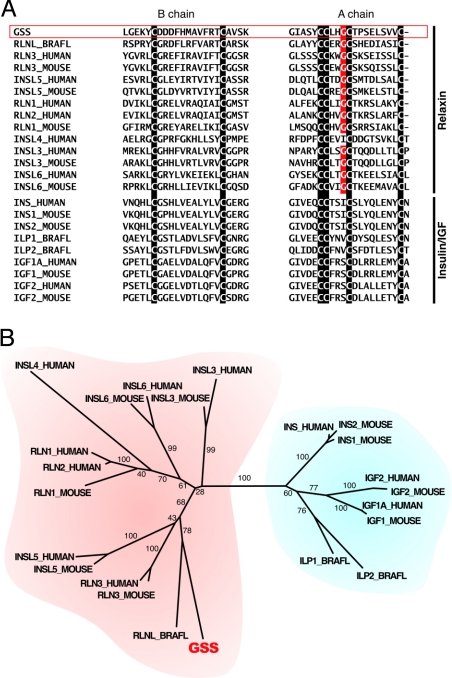
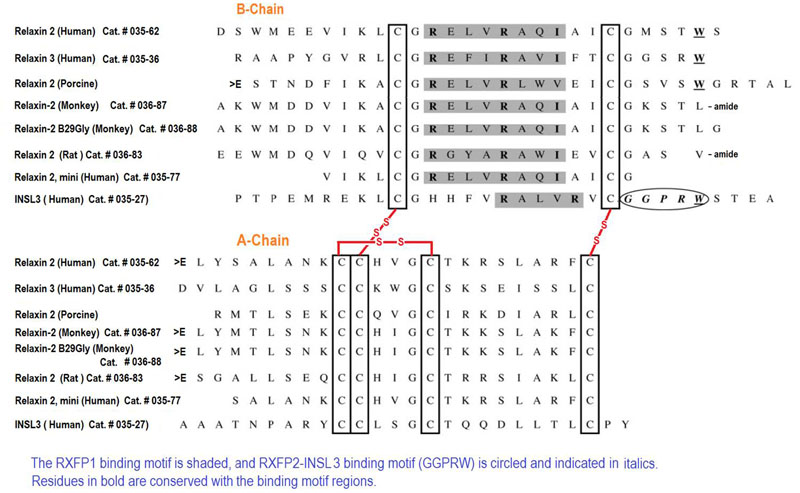
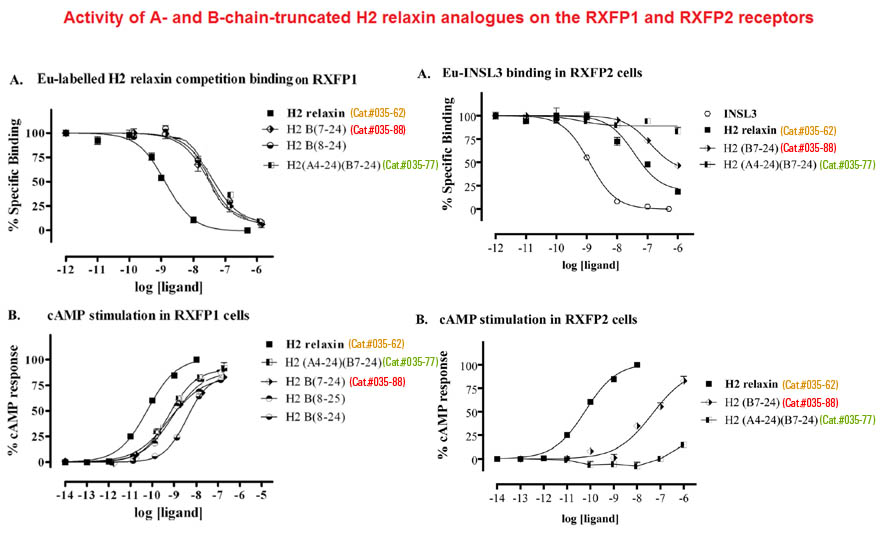
Human gene-2 (H2) relaxin is currently in Phase III clinical trials for the treatment of acute heart failure. It is a 53-amino acid insulin-like peptide comprising two chains and three disulfide bonds. It interacts with two of the relaxin family peptide (RXFP) receptors. Although its cognate receptor is RXFP1, it is also able to cross-react with RXFP2, the native receptor for a related peptide, insulin-like peptide 3. In order to understand the basis of this cross-reactivity, it is important to elucidate both binding and activation mechanisms of this peptide. The primary binding mechanism of this hormone has been extensively studied and well defined. H2 relaxin binds to the leucine-rich repeats of RXFP1 and RXFP2 using B-chain-specific residues. However, little is known about the secondary interaction that involves the A-chain of H2 relaxin and transmembrane exoloops of the receptors. We demonstrate here through extensive mutation of the A-chain that the secondary interaction between H2 relaxin and RXFP1 is not driven by any single amino acid, although residues Tyr-3, Leu-20, and Phe-23 appear to contribute. Interestingly, these same three residues are important drivers of the affinity and activity of H2 relaxin for RXFP2 with additional minor contributions from Lys-9, His-12, Lys-17, Arg-18, and Arg-22. Our results provide new insights into the mechanism of secondary activation interaction of RXFP1 and RXFP2 by H2 relaxin, leading to a potent and RXFP1-selective analog, H2:A(4-24)(F23A), which was tested in vitro and in vivo and found to significantly inhibit collagen deposition similar to native H2 relaxin.Chan LJ, Rosengren KJ, Layfield SL et al., J Biol Chem. 2012 Nov 30;287(49):41152-64. doi: 10.1074/jbc.M112.409284. Epub 2012 Sep 28.
Regulation of Sertoli cell number is a key event to determine normal spermatogenesis. We have previously shown that relaxin and its G-protein coupled receptor RXFP1 are expressed in rat Sertoli cells, and that relaxin stimulates Sertoli cell proliferation. This study examined the mechanisms underlying the mitogenic effect of relaxin in a primary culture of Sertoli cells removed from testes of immature rats. Stimulation with exogenous relaxin increased Sertoli cell number and the expression of the proliferating cell nuclear antigen (PCNA), but did not affect the mRNA level of the differentiation markers cadherins 1 and 2. Relaxin-induced Sertoli cell proliferation was blocked by inhibition of MEK/ERK1/2 or PI3K/AKT pathways, but not by inhibition of PKC or EGFR activity. Relaxin induced a rapid and transient activation of ERK1/2 phosphorylation, which was MEK and SRC-dependent, and involved upstream activation of G(i). AKT activation could be detected 5 min after relaxin stimulation, and was still detected after 24h of stimulation with relaxin. Relaxin-induced AKT phosphorylation was G(i)- but not PKA-dependent, and it was blocked by both PI3K and MEK inhibitors. In conclusion, the mitogenic effect of relaxin in Sertoli cell involves coupling to G(i) and activation of both MEK/ERK1/2 and PI3K/AKT pathways.Nascimento AR, Pimenta MT, Lucas TF, et al., Eur J Pharmacol. 2012 Sep 15;691(1-3):283-91. doi: 10.1016/j.ejphar.2012.07.021. Epub 2012 Jul 20.
Relaxin is a circulating hormone with functions in pregnancy, parturition, and other aspects of female reproduction. It is also secreted from the prostate gland into the seminal fluid; however, the role of relaxin in male reproduction is debated. Studies conducted in the past have suggested possible actions on human spermatozoa, but the data were contrasting. Here, we show that the relaxin receptor RXFP1 (Relaxin Family Peptide Receptor 1) is expressed in human spermatozoa, and it mainly localizes in the astrodome. In vitro studies on human sperm demonstrated that this hormone attenuates the natural decline in sperm motility and maintains higher mitochondrial activity and lower apoptosis level. Furthermore, relaxin induced an increase in sperm hyperactivation, intracellular calcium and cAMP, and acrosome reaction. These effects were abolished by the use of the specific anti-RXFP1 antibody. Relaxin concentrations were low in the blood (x¯ 6 SD, 0.16 6 0.03 nM) and very high in the seminal plasma (x¯ 6 SD, 10.3 6 4.0 nM), confirming its secretion mainly by the prostate. Taken together, these data demonstrate that relaxin influences positively many sperm functions linked to fertilizing ability, and it preserves sperm functionality, with possible practical value in assisted reproduction techniques.
This publication used Human relaxin-2 from Phoenix Pharmaceuticals for sperm function studies.
Ferlin A, Menegazzo M, Gianesello L, Selice R, Foresta C. Effect of relaxin on human sperm functions. J Androl. 2012;33(3):474-82.
Relaxin is a circulating hormone with functions in pregnancy, parturition, and other aspects of female reproduction. It is also secreted from the prostate gland into the seminal fluid; however, the role of relaxin in male reproduction is debated. Studies conducted in the past have suggested possible actions on human spermatozoa, but the data were contrasting. Here, we show that the relaxin receptor RXFP1 (Relaxin Family Peptide Receptor 1) is expressed in human spermatozoa, and it mainly localizes in the astrodome. In vitro studies on human sperm demonstrated that this hormone attenuates the natural decline in sperm motility and maintains higher mitochondrial activity and lower apoptosis level. Furthermore, relaxin induced an increase in sperm hyperactivation, intracellular calcium and cAMP, and acrosome reaction. These effects were abolished by the use of the specific anti-RXFP1 antibody. Relaxin concentrations were low in the blood (x ± SD, 0.16 ± 0.03 nM) and very high in the seminal plasma (x ± SD, 10.3 ± 4.0 nM), confirming its secretion mainly by the prostate. Taken together, these data demonstrate that relaxin influences positively many sperm functions linked to fertilizing ability, and it preserves sperm functionality, with possible practical value in assisted reproduction techniques.Ferlin A, Menegazzo M, Gianesello L, et al., J Androl. 2012 May-Jun;33(3):474-82. doi: 10.2164/jandrol.110.012625. Epub 2011 Sep 8.
H2 relaxin is a peptide hormone associated with a number of therapeutically relevant physiological effects, including regulation of collagen metabolism and multiple vascular control pathways. It is currently in phase III clinical trials for the treatment of acute heart failure due to its ability to induce vasodilation and influence renal function. It comprises 53 amino acids and is characterized by two separate polypeptide chains (A-B) that are cross-linked by three disulfide bonds. This size and complex structure represents a considerable challenge for the chemical synthesis of H2 relaxin, a major limiting factor for the exploration of modifications and derivatizations of this peptide, to optimize effect and drug-like characteristics. To address this issue, we describe the solid phase peptide synthesis and structural and functional evaluation of 24 analogues of H2 relaxin with truncations at the termini of its peptide chains. We show that it is possible to significantly truncate both the N and C termini of the B-chain while still retaining potent biological activity. This suggests that these regions are not critical for interactions with the H2 relaxin receptor, RXFP1. In contrast, truncations do reduce the activity of H2 relaxin for the related receptor RXFP2 by improving RXFP1 selectivity. In addition to new mechanistic insights into the function of H2 relaxin, this study identifies a critical active core with 38 amino acids. This minimized core shows similar antifibrotic activity as native H2 relaxin when tested in human BJ3 cells and thus represents an attractive receptor-selective lead for the development of novel relaxin therapeutics.Hossain MA, Rosengren KJ, Samuel CS et al., J Biol Chem. 2011 Oct 28;286(43):37555-65. doi: 10.1074/jbc.M111.282194. Epub 2011 Aug 30.
AIM: Relaxin is a polypeptide hormone involved in pregnancy and lactation. It is mainly secreted by the corpus luteum and placenta, but is expressed in a number of other tissues, including heart and brain. Within the brain, relaxin is expressed in the olfactory and limbic systems, the cortex and the hypothalamic arcuate nucleus (ARC). Its cognate receptor, relaxin family peptide receptor 1 (RXFP1), is also widely expressed in the brain, including the hypothalamic ARC and paraventricular nucleus (PVN), areas important in appetite regulation. The aim of this study was to investigate whether relaxin influences food intake through central hypothalamic circuits.METHODS: The human form of relaxin, human relaxin-2 (H2) was administered centrally and peripherally to male Wistar rats and food intake measured. Behaviour was also assessed.RESULTS: Intracerebroventricular (ICV) administration of H2 significantly decreased 1-h food intake in the early dark phase [2.95 ± 0.45 g (saline) vs. 0.95 ± 0.18 g (180 pmol H2), p < 0.001]. ICV administration of H2 decreased feeding behaviour and increased grooming and headdown behaviour. Intraparaventricular injections of H2 significantly decreased 1-h food intake in the early dark phase [3.13 ± 0.35 g (saline) vs. 1.35 ± 0.33 g (18 pmol H2), p < 0.01, 1.61 ± 0.31 g (180 pmol H2), p < 0.05 and 1.23 ± 0.32 g (540 pmol H2), p < 0.001]. Intraperitoneal (IP) administration of H2 significantly decreased 1-h food intake in the early dark phase [4.63 ± 0.46 g (vehicle) vs. 3.08 ± 0.15 g (66 nmol H2), p < 0.01, 3.00 ± 0.17 g (200 nmol H2), p < 0.01 and 2.26 ± 0.36 g (660 nmol H2), p < 0.001].CONCLUSIONS: Central and peripheral administration of H2 reduces the food intake in rats. This effect may be mediated via the PVN and/or other brain regions.McGowan BM, Minnion JS, Murphy KG et al., Diabetes Obes Metab. 2010 Dec;12(12):1090-6. doi: 10.1111/j.1463-1326.2010.01302.x.
BACKGROUND: Relaxin is the endogenous ligand of the G-protein coupled receptor RXFP1, previously known as LGR7. In humans relaxin can also activate, but with lower affinity, the closely related receptor for the insulin-like peptide from Leydig cells, RXFP2, previously known as LGR8. The lack of relaxin impairs male fertility but the precise distribution and the function of relaxin receptors in the male reproductive tract is not known. We investigated the distribution of Rxfp1 and Rxfp2 in the reproductive tract of the male rat and the function of relaxin in the vas deferens, a tissue with high expression of both receptors.METHODS: The presence of mRNA for Rxfp1 and Rxfp2 was investigated in testes, cultured Sertoli cells, epididymis, vas deferens, seminal vesicle, prostate, and spermatozoa by RT-PCR and Southern blot. Protein expression in the testis, vas deferens, primary culture of Sertoli cells, and spermatozoa was assessed by immunohistochemistry and immunofluorescence. The role of relaxin in the vas deferens was evaluated by contractility studies and radioimmunoassay of cAMP production. The effect of relaxin on mRNA levels for metalloproteinase-7 was measured by Northern blot.RESULTS: Transcripts for Rxfp1 and Rxfp2 were present in almost all parts of the male reproductive tract, with high levels in testis and vas deferens. Both receptors were immunolocalized in late stage germ cells but not in mature spermatozoa, although mRNAs for both receptors were also present in mature spermatozoa. Rxfp1 but not Rxfp2 was detected in cultured Sertoli cells. Strong immunostaining for Rxfp1 and Rxfp2 was seen in muscular and epithelial layers of the vas deferens and in arteriolar walls. Relaxin did not affect contractility and cyclic AMP production of the vas deferens, but increased the levels of mRNA for metalloproteinase-7.CONCLUSION: Rxfp1 and Rxfp2 are widely and similarly distributed throughout the male reproductive tract. Our results suggest that Rxfp1 on spermatids and Sertoli cells may be important in spermatogenesis. Relaxin in the vas deferens does not affect contractility, but may affect vascular compliance and collagen and matrix remodeling.This Publication used LGR7 and LGR8 antibodies from Phoenix Pharmaceuticals for IHC staining.
Filonzi M, Cardoso LC, Pimenta MT, et al. Reprod Biol Endocrinol. 2007;5:29.
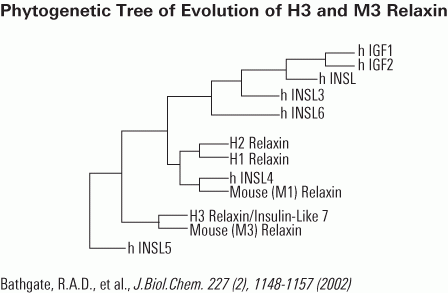
In humans, the relaxin hormone family includes H1, H2 and H3 isoforms and insulin-like peptides 3 to 6. The ever-increasing interest in relaxin as potential new drug requires reliable methods to compare bioactivity of different relaxins. The existing bioassays include in vivo or ex vivo methods evaluating the organ-specific responses to relaxin and in vitro methods based on measurement of cAMP increase in relaxin receptor-bearing cells. We previously demonstrated that relaxin dose-dependently inhibits platelet aggregation. On this basis, we have developed a simple, reliable bioassay for relaxin used to compare purified porcine relaxin, assumed as reference standard, with two recombinant human H2 relaxins, H3 relaxin, insulin-like peptides 3 and 5. Pre-incubation of platelets with relaxins (3, 10, 30,100, 300 ng/ml; 10 min.) caused the inhibition of ADP-induced platelet aggregation. Within the 10-100 ng/ml range, porcine relaxin showed the highest effects and a nearly linear dose-response correlation. Lower peptide concentrations were ineffective, as were insulin-like peptides 3 and 5 at any concentration assayed. Platelet inhibition was mediated by specific RXFP1 relaxin receptor and cGMP, whose intracellular levels dose-dependently increased upon relaxin. For comparison, we stimulated THP-1 cells, a relaxin receptor-bearing cell line, with porcine relaxin, human H2 and H3 relaxins at the above concentrations (15 min.). We observed a dose-related increase of intracellular cAMP similar to the trend of platelet inhibition. Insulin like peptide 5 was ineffective. In conclusion, this study shows that inhibition of platelet aggregation may be used to assess bioactivity of relaxin preparations for experimental and clinical purposes.
This publication used human relaxin-2 peptide from Phoenix Pharmaceuticals for ADP-induced platelet aggregation study.
Bani D, Nistri S, Cinci L, et al. A novel, simple bioactivity assay for relaxin based on inhibition of platelet aggregation. Regul Pept. 2007;144(1-3):10-6.
AIM: To investigate the severity of acute pancreatitis (AP) is associated to the intensity of leukocyte activation, inflammatory up-regulation and microcirculatory disruption associated to ischemia-reperfusion injury. Microvascular integrity and inhibition of pro-inflammatory mediators are key-factors in the evolution of AP. Relaxin is an insulin-like hormone that has been attributed vasorelaxant properties via the nitric oxide pathway while behaving as a glucocorticoid receptor agonist.METHODS: AP was induced by the bilio-pancreatic duct-outlet-exclusion closed-duodenal-loops model. Treatment with relaxin was done at different time-points. Nitric oxide synthase inhibition by L-NAME and glucocorticoid receptor (GR) blockage by mifepristone was considered. AP severity was assessed by biochemical and histopathological analyses.RESULTS: Treatment with relaxin reduced serum amylase, lipase, C-reactive protein, IL-6, IL-10, hsp72, LDH and 8-isoprostane as well as pancreatic and lung myeloperoxidase. Acinar and fat necrosis, hemorrhage and neutrophil infiltrate were also decreased. ATP depletion and ADP/ATP ratio were reduced while caspases 2-3-8 and 9 activities were increased. L-NAME and mifepristone decreased the efficiency of relaxin.CONCLUSION: Relaxin resulted beneficial in the treatment of AP combining the properties of a GR agonist while preserving the microcirculation and favoring apoptosis over necrosis.Cosen-Binker LI, Binker MG, Cosen R et al., World J Gastroenterol. 2006 Mar 14;12(10):1558-68.
Relaxin peptides are important hormones for the regulation of reproductive tissue remodeling and the renal cardiovascular system during pregnancy. Recent studies demonstrated that two of the seven human relaxin family peptides, relaxin H2 (RLN2) and INSL3, signal exclusively through leucine-rich repeat-containing G protein-coupled receptors, LGR7 and LGR8. Although it was well characterized that an RXXXRXXI motif at the RLN2 B chain confers receptor activation activity, it is not clear what roles RLN2 A chain plays in receptor interaction. Analyses of relaxin family genes on syntenic regions of model tetrapods showed that the A chain of RLN2 orthologs exhibited a greater sequence divergence as compared with the receptor-binding domain-containing B chain, foreshadowing a potential role in receptorinteractions; hence, defining receptor selectivity in this fast evolving peptide hormone. To test our hypothesis that select residues in the human RLN2 A chain play key roles in receptor interaction, we studied mutant peptides with residue substitution(s) in the A chain. Here, we showed that alanine substitution at the A16 and A17 positions enhances LGR8-activation activity of RLN2, whereas mutation at the A22-23 region (RLN2A22-23) ablates LGR8, but not LGR7, activation activity. In addition, we demonstrated that the functional characteristics of the RLN2A22-23 mutant are mainly attributed to modifications at the PheA23 position. Taken together, our studies indicated that ThrA16, LysA17, and PheA23 constitute part of the receptor-binding interface of human RLN2, and that modification of these residues has led to the generation of novel human RLN2 analogs that would allow selective activation of human LGR7, but not LGR8, in vivo.
The publication used human relaxin 2 peptide tracer (Cat.#T-035-62) for receptor binding experiments
Park JI, Semyonov J, Yi W, Chang CL, Hsu SY. J Biol Chem. 2008;283(46):32099-109.
The omnipresent 6-kDa polypeptide relaxin (RLX) is emerging as a multifunctional endocrine and paracrine factor in a broad range of target tissues including cardiovascular tissues. To explore the pathophysiological roles of RLX in ischemic cardiovascular diseases, we studied the changes in RLX mRNA level in the myocardium and the effect of RLX supplements in rats with isoproterenol (ISO)-induced myocardial injury. In ISO-treated rats, RLX levels in myocardia and plasma increased 3.7- and 6.9-fold, respectively (P<0.01), the mRNA level increased significantly in myocardia compared with controls. Co-administration of RLX (0.2 and 2.0 microg/kg/d) and ISO increased left-ventricular pressure development and decreased left ventricular end-diastolic pressure (LVDEP) (all P<0.01). Malondialdehyde content in myocardia and lactate dehydrogenase and creatine phosphokinase activities in plasma in RLX-treated rats decreased markedly compared with that in ISO-treated alone rats (P<0.01 or P<0.05). In the high-dose RLX group, fibroblastic hyperplasia was relieved in myocardia, hydroxyproline level was lower, by 33% (P<0.05), and endothelin content in plasma was lower, by 31% (P<0.01) than in the ISO-alone group. Compared with control group, any indexes in sham rats treated with high-dose RLX were unaltered (all P>0.05). These results showed an up-regulation of myocardial RLX during ISO-induced myocardial ischemia injury and the protective effect of RLX on ISO-induced cardiac inhibition and fibrosis, which suggests that RLX could be an endogenous cardioprotective factor in ischemic heart diseases.
Zhang J, Qi YF, Geng B, et al., Peptides. 2005 Sep;26(9):1632-9.
Title
Lead Author
Publication
Institution(s)
Bioactivities
Relaxin improves TNF-α-induced endothelial dysfunction: the role of glucocorticoid receptor and phosphatidylinositol 3-kinase signaling
Dschietzig T
Cardiovasc Res. 2012 ;95(1):97-107. doi: 10.1093/cvr/cvs149
Immundiagnostik AG, Stubenwaldallee 8a, Bensheim, Germany.
improves endothelial function by inhibiting vascular inflammation
Relaxin 2 is functional at the ocular surface and promotes corneal wound healing
Hampel U
Invest Ophthalmo Vis Sci. 2012 ;53(12):7780-7790. doi: 10.1167/iovs.12-10714
Department of Anatomy II, Friedrich Alexander University of Erlangen-Nürnberg, Erlangen, Germany.
Improves cell proliferation, migration and re-epithelialization of corncal wounds
Relaxin reduces xenograft tumor growth of human MDA-MB-231 breast cancer cells
Y Radestock
Breast Cancer Research 2008, 10:1-15. doi:10.1186/bcr2136
Clinics of General, Visceral and Vascular Surgery, Magdeburger Str. 18, Martin-Luther-University Halle Wittenberg, 06097 Halle, Germany.
increases cell motility dose-dependently
In Vitro Pharmacological Characterization of RXFP3 Allosterism: An Example of Probe Dependency
Alvarez-Jaimes L
PLoS One. 2012;7(2):e30792. doi: 10.1371/journal.pone.0030792. Epub 2012 Feb 7.
Janssen Pharmaceutical Companies of Johnson & Johnson, San Diego, California, United States of America
cAMP accumulation
Effect of Relaxin on Human Sperm Functions and Fertilizing Ability
Ferlin A
Journal of Andrology, (33), 3, 474-80, 2012
University of padova, Padova, Italy
increase in sperm hyperactivation, intracellular calcium and cAMP, and induce acrosome reaction
Effect of relaxin on myocardial ischemia injury induced by isoproterenol
Zhang J
Peptides. 2005 Sep;26(9):1632-1639
Institute of Cardiovascular Research, Peking University , China
Protect from myocardial ischemia injury and fibrosis
Intracellular signaling pathways involved in the relaxin-induced proliferation of rat Sertoli cells.
Nascimento A R
Eur J Pharmacol. 2012 Sep 15;691(1-3):283-91. doi: 10.1016/j.ejphar.2012.07.021
Universidade Federal de São Paulo, São Paulo, Brazil.
Induced Sertoli cell proliferation, and activation the activity of ERK1/2 phosphorylation and G(i)
Relaxin prevents the development of severe acute pancreatitis
Laura Iris Cosen-Binker
World J Gastroenterol 2006 March 14;12(10):1558-1568;
RHC-LICB Medical Research Institute-Buenos Aires, Argentina
prevents the development of severe acute pancreatitis
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 035-62 | Relaxin 2 (Human) | 20 µg | $247 |
| 035-62A | Relaxin 2 (Human) | 100 µg | $444 |
| T-035-62 | Relaxin 2 (Human) - I-125 Labeled | 10 µCi | $1322 |
| 036-86 | Relaxin (Mouse) | 100 µg | $434 |
| 036-84 | Relaxin (Rat) | 100 µg | $434 |
| FC5-035-62 | Relaxin 2 (Human) - Cy5 Labeled | 1 nmol | $555 |
| 035-83 | Relaxin 2, Extended B-Chain (1-31) (Human) | 100 µg | $382 |
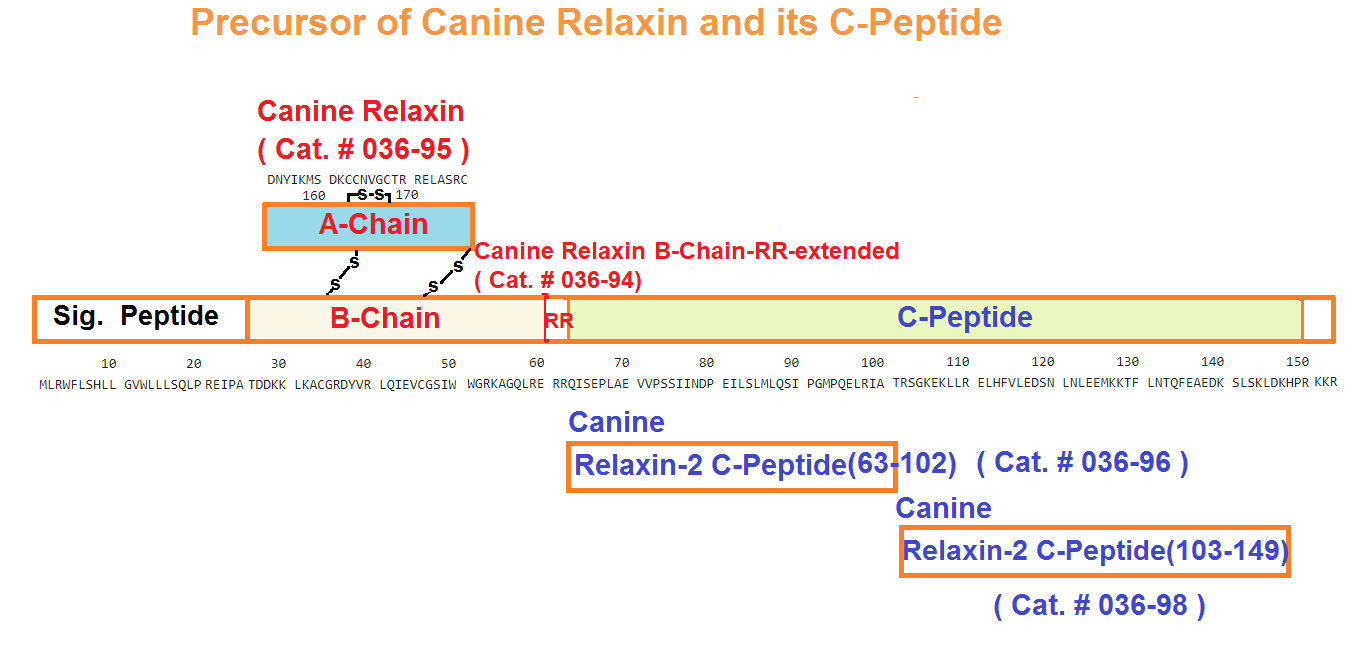
| 036-94 | Canine Relaxin with B-chain-RR-extended | 100 μg | $368 |
| 036-89 | RLN2-2 / Relaxin-2 Isoform 2 (Human) | 100 µg | $428 |
| 036-51 | RXFP3 specific agonist [G(B24)S]R3/I5 | 100 µg | $428 |
Social Network Confirmation