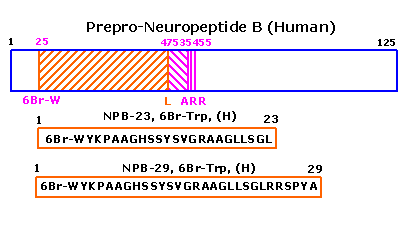
  |
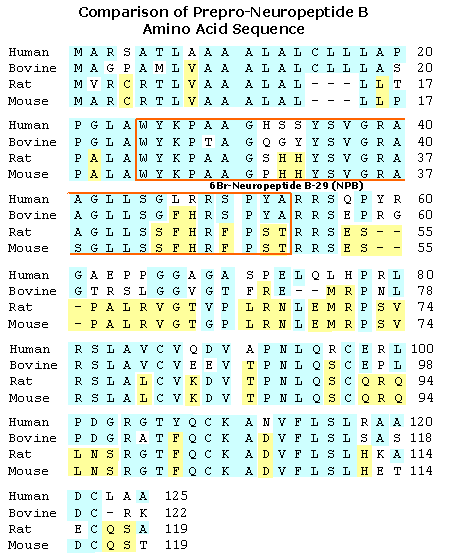  |
GPR7 and GPR8 are two structurally related orphan G protein-coupled receptors, presenting high similarities with opioid and somatostatin receptors. Two peptides, L8 and L8C, derived from a larger precursor, were recently described as natural ligands for GPR8 (Mori, M., Shimomura, Y., Harada, M., Kurihara, M., Kitada, C., Asami, T., Matsumoto, Y., Adachi, Y., Watanabe, T., Sugo, T., and Abe, M. (December, 27, 2001) World Patent Cooperation Treaty, Patent Application WO 01/98494A1). L8 is a 23-amino acid peptide, whereas L8C is the same peptide with a C terminus extension of 7 amino acids, running through a dibasic motif of proteolytic processing. Using as a query the amino acid sequence of the L8 peptide, we have identified in DNA databases a human gene predicted to encode related peptides and its mouse ortholog. By analogy with L8 and L8C, two peptides, named L7 and L7C could result from the processing of a 125-amino acid human precursor through the alternative usage of a dibasic amino acid motif. The activity of these four peptides was investigated on GPR7 and GPR8. In binding assays, L7, L7C, L8, and L8C were found to bind with low nanomolar affinities to the GPR7 and GPR8 receptors expressed in Chinese hamster ovary (CHO)-K1 cells. They inhibited forskolin-stimulated cAMP accumulation through a pertussis toxin-sensitive mechanism. The tissue distribution of prepro-L7 (ppL7) and prepro-L8 (ppL8) was investigated by reverse transcription-PCR. Abundant ppL7 transcripts were found throughout the brain as well as in spinal cord, spleen, testis, and placenta; ppL8 transcripts displayed a more restricted distribution in brain, with high levels in substantia nigra, but were more abundant in peripheral tissues. The ppL7 and ppL8 genes therefore encode the precursors of a class of peptide ligands, active on two receptor subtypes, GPR7 and GPR8. The distinct tissue distribution of the receptor and peptide precursors suggest that each ligand and receptor has partially overlapping but also specific roles in this signaling system.
Brezillon S, Lannoy V, Franssen JD, et al. Identification of natural ligands for the orphan G protein-coupled receptors GPR7 and GPR8. J Biol Chem. 2003;278(2):776-83.
Gold-thioglucose (GTG) induces lesions in the ventromedial nucleus of the hypothalamus, resulting in hyperphagia and obesity. To identify genes involved in the hypothalamic regulation of energy homeostasis, we used a screen for genes that are dysregulated in GTG-induced obese mice. We found that GPR7, the endogenous G protein-coupled receptor for the recently identified ligands neuropeptide B and neuropeptide W, was down-regulated in hypothalamus after GTG treatment. Here we show that male GPR7-/- mice develop an adult-onset obese phenotype that progressively worsens with age and was greatly exacerbated when animals are fed a high-fat diet. GPR7-/- male mice were hyperphagic and had decreased energy expenditure and locomotor activity. Plasma levels of glucose, leptin, and insulin were also elevated in these mice. GPR7-/- male mice had decreased hypothalamic neuropeptide Y RNA levels and increased proopiomelanocortin RNA levels, a set of effects opposite to those evident in ob/ob mice. Furthermore, ob/ob GPR7-/- and Ay/a GPR7-/- double mutant male mice had an increased body weight compared with normal ob/ob or Ay/a male mice, suggesting that the obesity of GPR7-/- mice is independent of leptin and melanocortin signaling. Female mice did not show any significant weight increase or associated metabolic defects. These data suggest a potential role for GPR7 and its endogenous ligands, neuropeptide B and neuropeptide W, in regulating energy homeostasis independent of leptin and melanocortin signaling in a sexually dimorphic manner.
Ishii M, Fei H, Friedman JM. Targeted disruption of GPR7, the endogenous receptor for neuropeptides B and W, leads to metabolic defects and adult-onset obesity. Proc Natl Acad Sci USA. 2003;100(18):10540-5.
GPR7 and GPR8 are orphan G protein-coupled receptors that are highly similar to each other. These receptors are expressed predominantly in brain, suggesting roles in central nervous system function. We have purified an endogenous peptide ligand for GPR7 from bovine hypothalamus extracts. This peptide, termed neuropeptide B (NPB), has a C-6-brominated tryptophan residue at the N terminus. It binds and activates human GPR7 or GPR8 with median effective concentrations (EC(50)) of 0.23 nM and 15.8 nM, respectively. In situ hybridization shows distinct localizations of the prepro-NPB mRNA in mouse brain, i.e., in paraventricular hypothalamic nucleus, hippocampus, and several nuclei in midbrain and brainstem. Intracerebroventricular (i.c.v.) injection of NPB in mice induces hyperphagia during the first 2 h, followed by hypophagia. Intracerebroventricular injection of NPB produces analgesia to s.c. formalin injection in rats. Through EST database searches, we identified a putative paralogous peptide. This peptide, termed neuropeptide W (NPW), also has an N-terminal tryptophan residue. Synthetic human NPW binds and activates human GPR7 or GPR8 with EC(50) values of 0.56 nM and 0.51 nM, respectively. The expression of NPW mRNA in mouse brain is confined to specific nuclei in midbrain and brainstem. These findings suggest diverse physiological functions of NPB and NPW in the central nervous system, acting as endogenous ligands on GPR7 andor GPR8.
Tanaka H, Yoshida T, Miyamoto N, et al. Characterization of a family of endogenous neuropeptide ligands for the G protein-coupled receptors GPR7 and GPR8. Proc Natl Acad Sci USA. 2003;100(10):6251-6.
We isolated a novel gene in a search of the Celera data base and found that it encoded a peptidic ligand for a G protein-coupled receptor, GPR7 (O'Dowd, B. F., Scheideler, M. A., Nguyen, T., Cheng, R., Rasmussen, J. S., Marchese, A., Zastawny, R., Heng, H. H., Tsui, L. C., Shi, X., Asa, S., Puy, L., and George, S. R. (1995) Genomics 28, 84-91; Lee, D. K., Nguyen, T., Porter, C. A., Cheng, R., George, S. R., and O'Dowd, B. F. (1999) Mol. Brain Res. 71, 96-103). The expression of this gene was detected in various tissues in rats, including the lymphoid organs, central nervous system, mammary glands, and uterus. GPR7 mRNA was mainly detected in the central nervous system and uterus. In situ hybridization showed that the gene encoding the GPR7 ligand was expressed in the hypothalamus and hippocampus of rats. To determine the molecular structure of the endogenous GPR7 ligand, we purified it from bovine hypothalamic tissue extracts on the basis of cAMP production-inhibitory activity to cells expressing GPR7. Through structural analyses, we found that the purified endogenous ligand was a peptide with 29 amino acid residues and that it was uniquely modified with bromine. We subsequently determined that the C-6 position of the indole moiety in the N-terminal Trp was brominated. We believe this is the first report on a neuropeptide modified with bromine and have hence named it neuropeptide B. In in vitro assays, bromination did not influence the binding of neuropeptide B to the receptor.
Fujii R, Yoshida H, Fukusumi S, et al. Identification of a neuropeptide modified with bromine as an endogenous ligand for GPR7. J Biol Chem. 2002;277(37):34010-6.
The structurally related orphan G-protein-coupled receptors GPR7 and GPR8 are expressed in the central nervous system, and their ligands have not been identified. Here, we report the identification of the endogenous ligand for both of these receptors. We purified the peptide ligand from porcine hypothalamus using stable Chinese hamster ovary cell lines expressing human GPR8 and cloned the cDNA encoding its precursor protein. The cDNA encodes two forms of the peptide ligand with lengths of 23 and 30 amino acid residues as mature peptides. We designated the two ligands neuropeptide W-23 (NPW23) and neuropeptide W-30 (NPW30). The amino acid sequence of NPW23 is completely identical to that of the N-terminal 23 residues of NPW30. Synthetic NPW23 and NPW30 activated and bound to both GPR7 and GPR8 at similar effective doses. Intracerebroventricular administration of NPW23 in rats increased food intake and stimulated prolactin release. These findings indicate that neuropeptide W is the endogenous ligand for both GPR7 and GPR8 and acts as a mediator of the central control of feeding and the neuroendocrine system.
Migeotte I, Riboldi E, Franssen JD, et al. Identification and characterization of an endogenous chemotactic ligand specific for FPRL2. J Exp Med. 2005;201(1):83-93.
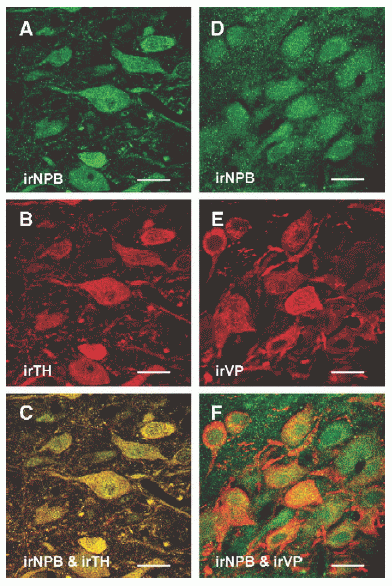 |
Confocal images of rat midbrain (A– C) and hypothalamic sections (D–F) double labeled with neuropeptide B antiserum (NPB, green image) and tyrosine hydroxylase (TH) or vasopressin (VP) antibody. In the substantia nigra pars compacta, all the irNPB cells (A) are TH positive (B). (C) A merged image of panels A and B. In the supraoptic nucleus, some of the VP-neurons (E) express NPB immunoreactivity (D). (F) A merged image of panels D and E, where co-expression of NPB and VP immunoreactivity is clearly detected in several neurons. Scale bar: 20 µm.
|
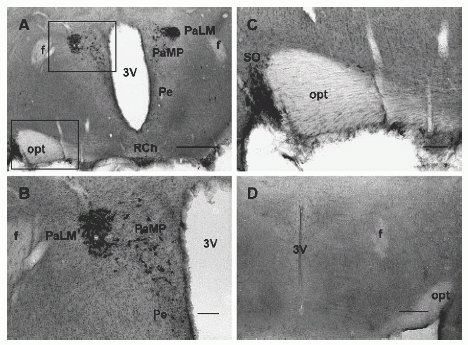 |
Photomicrographs of sections through the rat hypothalamus labeled with or without NPB antiserum. (A) Strongly labeled neurons are seen in the lateromagnocellular part (PaLM) and medial parvocellular part (PaMP) of the paraventricular hypothalamic nucleus and in the supraoptic nucleus (SO). Scattered cells are present in the periventricular hypothalamic nucleus (Pe) and retrochiasmatic nucleus (RCh). (B) A higher magnification of the top area outlined in panel A where many immunoreactive cells from PaLM and PaMP are clearly visible; smaller, positively labeled cells are seen in the Pe. (C) A higher magnification of the bottom area outlined in panel A, where labeled cells are present in SO. (D) A hypothalamic section processed without NPB antiserum; immunoreactivity is not noted in this section. Abbreviations: f, fornix; opt, optic tract; 3V, 3rd ventricle. Scale bar: panels A and D, 250 µm; panels B and C, 100 µm. |
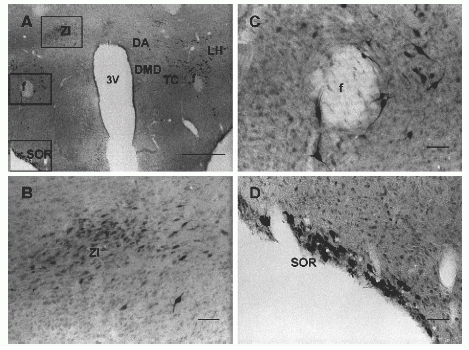 |
Photomicrographs of sections through the caudal rat hypothalamus labeled with NPB antiserum. (A) Intensely labeled cells are seen in the supraoptic retrochiasmatic nucleus (SOR); immunoreactive cells were also present in zona incerta (ZI), perifornical area, lateral hypothalamic area (LH), dorsal hypothalamic area (DA), dorsomedial hypothalamic nucleus dorsal part (DMD), and tuber cinereum area (TC). (B) A higher magnification of the top area outlined in panel A, where moderately labeled irNPB neurons are present in the zona incerta (ZI). (C) A higher magnification of the area around the fornix (f) in panel A, where immunoreactive cells are seen surrounding the fornix. (D) A higher magnification of the bottom area outlined in panel Awhere strongly labeled neurons are present in SOR. Abbreviations: 3V, 3rd ventricle. Scale bar: panel A, 250 µm; panels B– D, 50 µm. |
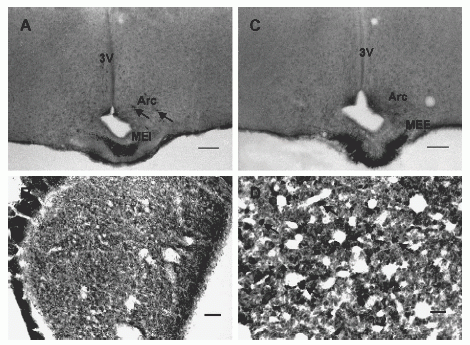 |
Photomicrographs of sections through the rat hypothalamus and pituitary labeled with NPB antiserum. (A) Intensely labeled fibers are present in the internal layer of the median eminence (MEI) and few small cells are present in the arcuate nucleus (Arc; arrows). (B) A section of posterior pituitary where NPB-immunoreactive cell processes/terminals and occasionally small cells are noted. (C) Intensely labeled fibers are noted in the external layer of the median eminence (MEE). (D) A section of anterior pituitary where many small NPB-labeled cells are seen; arrows point to representative cells. Abbreviations: 3V, 3rd ventricle. Scale bar: panels A and C, 250 µm; panels B and D, 100 µm. |
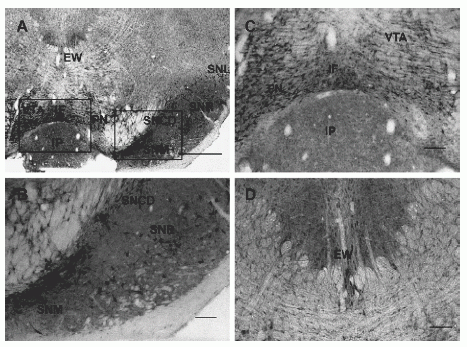 |
Photomicrographs of sections through the rat midbrain labeled with NPB antiserum. (A) Immunoreactive cells are present in Edinger–Westphal nucleus (EW), substantia nigra: compact part, dorsal tier (SNCD), reticular part (SNR), lateral part (SNL) and medial part (SNM), paranigral nucleus (PN), ventral tegmental area (VTA), and interfascicular nucleus (IF). (B) A higher magnification of the area outlined in panel A showing many immunoreactive neurons within the substantia nigra; there is a higher density of cells in the SNM and SNCD as compared to the SNR. (C) A higher magnification of the area outlined in panel Awhere irNPB neurons are seen in the IF, PN, and VTA. (D) Intensely labeled neurons are present in the EW. Abbreviations: IP, interpeduncular nucleus. Scale bar: panel A, 250 µm; panels B– D, 100 µm. |
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| 005-49 | prepro-Neuropeptide B (NPB-29) (56-81) (Human) | 100 µg | $242 |
| B-005-49 | prepro-Neuropeptide B (NPB-29) (56-81) (Human) - Biotin Labeled | 100 µg | $306 |
| EK-005-49 | prepro-Neuropeptide B (NPB-29) (56-81) (Human) - EIA Kit | 96 wells | $610 |
| 005-53 | [Des-Br]-Neuropeptide B-23 (NPB-23) / L7 (Human) | 100 µg | $167 |
| FG-005-53A | [Des-Br]-Neuropeptide B-23 (NPB-23) / L7 (Human) - FAM Labeled | 1 nmol | $382 |
| T-005-53 | [Des-Br]-Neuropeptide B-23 (NPB-23) / L7 (Human) - I-125 Labeled | 10 µCi | $1082 |
| FR-005-53 | [Des-Br]-Neuropeptide B-23 (NPB-23) / L7 (Human) - Rhodamine Labeled | 1 nmol | $444 |
| 005-54 | Neuropeptide B-29 (NPB-29) (Bovine) | 100 µg | $267 |
| 005-55 | [Des-Br]-Neuropeptide B-29 (NPB-29) (Bovine) | 100 µg | $202 |
| 005-67 | [Des-Br-D-Trp]-Neuropeptide B-29 (NPB-29) (Bovine) | 100 µg | $202 |
Social Network Confirmation