The aggregation and deposition of amyloid-β (Aβ) peptides are believed to be central events in the pathogenesis of Alzheimer's disease (AD). Inoculation of brain homogenates containing Aβ aggregates into susceptible transgenic mice accelerated Aβ deposition, suggesting that Aβ aggregates are capable of self-propagation and hence might be prions. Recently, we demonstrated that Aβ deposition can be monitored in live mice using bioluminescence imaging (BLI). Here, we use BLI to probe the ability of Aβ aggregates to self-propagate following inoculation into bigenic mice. We report compelling evidence that Aβ aggregates are prions by demonstrating widespread cerebral β-amyloidosis induced by inoculation of either purified Aβ aggregates derived from brain or aggregates composed of synthetic Aβ. Although synthetic Aβ aggregates were sufficient to induce Aβ deposition in vivo, they exhibited lower specific biological activity compared with brain-derived Aβ aggregates. Our results create an experimental paradigm that should lead to identification of self-propagating Aβ conformations, which could represent novel targets for interrupting the spread of Aβ deposition in AD patients.
Stöhr J, Watts JC, Mensinger ZL, et al. Purified and synthetic Alzheimer's amyloid beta (Aβ) prions. Proc Natl Acad Sci USA. 2012;109(27):11025-30.
In Alzheimer's disease (AD), dementia severity correlates strongly with decreased synapse density in hippocampus and cortex. Numerous studies report that hippocampal long-term potentiation (LTP) can be inhibited by soluble oligomers of amyloid β-protein (Aβ), but the synaptic elements that mediate this effect remain unclear. We examined field EPSPs and whole-cell recordings in wild-type mouse hippocampal slices. Soluble Aβ oligomers from three distinct sources (cultured cells, AD cortex, or synthetic peptide, Aβ S26C dimers) inhibited LTP, and this was prevented by the selective NR2B inhibitors ifenprodil and Ro 25-6981. Soluble Aβ enhanced NR2B-mediated NMDA currents and extrasynaptic responses; these effects were mimicked by the glutamate reuptake inhibitor dl-threo-β-benzyloxyaspartic acid. Downstream, an Aβ-mediated rise in p38 mitogen-activated protein kinase (MAPK) activation was followed by downregulation of cAMP response element-binding protein, and LTP impairment was prevented by inhibitors of p38 MAPK or calpain. Thus, soluble Aβ oligomers at low nanomolar levels present in AD brain increase activation of extrasynaptic NR2B-containing receptors, thereby impairing synaptic plasticity.
Li S, Jin M, Koeglsperger T, Shepardson N, Shankar G, Selkoe D. Soluble Aβ oligomers inhibit long-term potentiation through a mechanism involving excessive activation of extrasynaptic NR2B-containing NMDA receptors. The Journal of neuroscience?: the official journal of the Society for Neuroscience. 2011;31(18):6627-6638. doi:10.1523/JNEUROSCI.0203-11.2011.
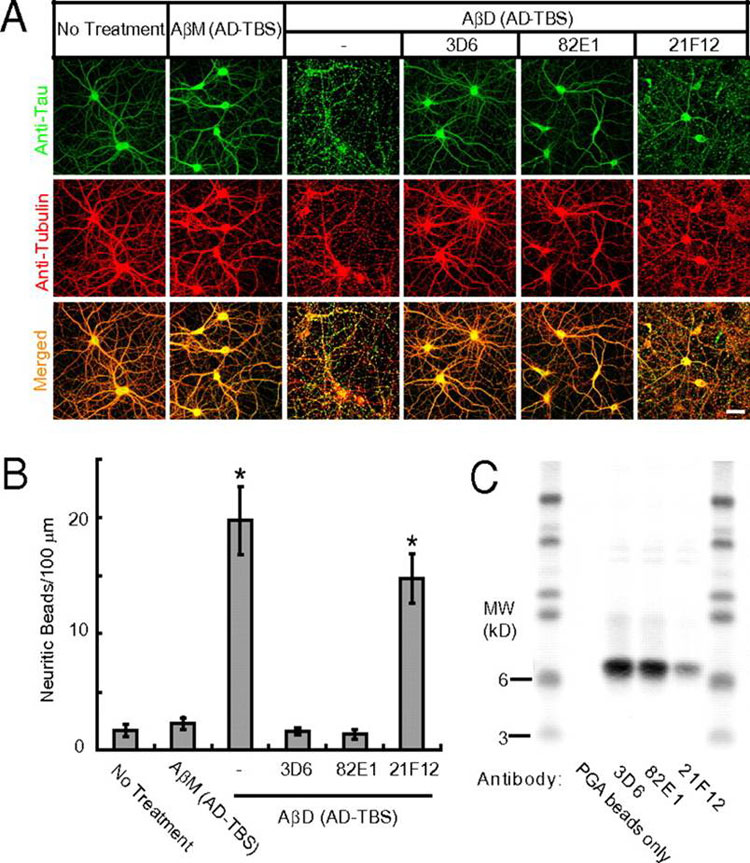
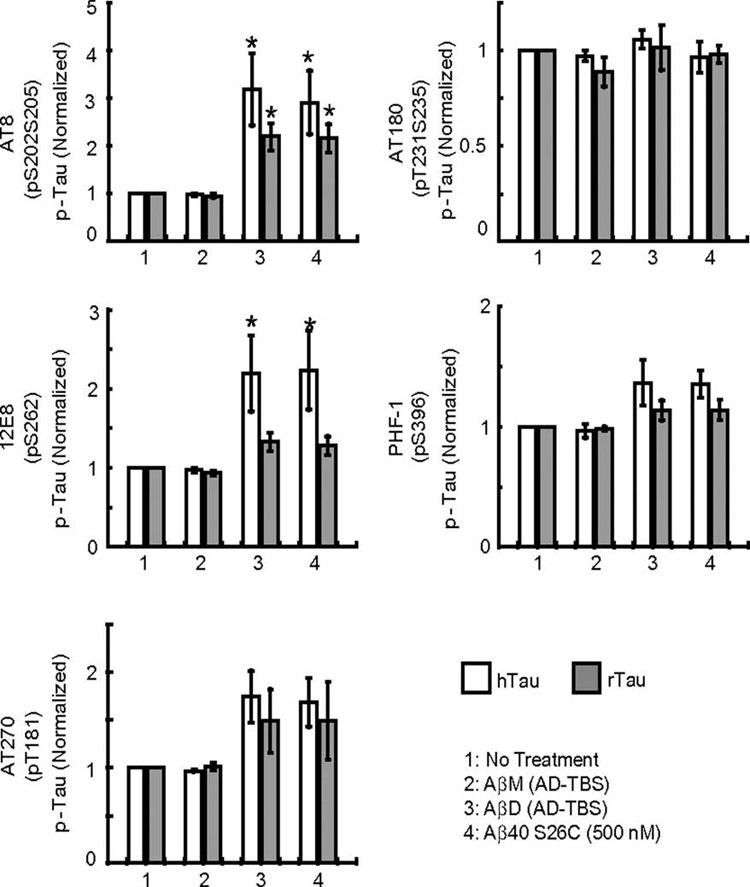
Alzheimer disease is a major cause of cognitive failure, and a pathogenically related but more subtle process accounts for many cases of mild memory symptoms in older humans. Insoluble fibrillar plaques of amyloid β-proteins (Aβ) and neurofibrillary deposits of hyperphosphorylated tau proteins are the diagnostic lesions of AD, but their temporal mechanistic relationship has long been debated. The recent recognition that small, diffusible oligomers may be the principal bioactive form of Aβ raises the key question of whether these are sufficient to initiate cytoskeletal change and neurite degeneration. A few studies have examined the effects of oligomers of synthetic Aβ peptides of one defined length at supraphysiological concentrations, but the existence of such assemblies in the AD brain is not established. Here, we isolated Aβ dimers, the most abundant form of soluble oligomer detectable in the human brain, from the cortices of typical AD subjects and found that at subnanomolar concentrations, they first induced hyperphosphorylation of tau at AD-relevant epitopes in hippocampal neurons and then disrupted the microtubule cytoskeleton and caused neuritic degeneration, all in the absence of amyloid fibrils. Application of pure, synthetic dimers confirmed the effects of the natural AD dimers, although the former were far less potent. Knocking down endogenous tau fully prevented the neuritic changes, whereas overexpressing human tau accelerated them. Coadministering Aβ N-terminal antibodies neutralized the cytoskeletal disruption. We conclude that natural dimers isolated from the AD brain are sufficient to potently induce AD-type tau phosphorylation and then neuritic dystrophy, but passive immunotherapy mitigates this.
Jin M, Shepardson N, Yang T, Chen G, Walsh D, Selkoe DJ. Soluble amyloid beta-protein dimers isolated from Alzheimer cortex directly induce Tau hyperphosphorylation and neuritic degeneration. Proc Natl Acad Sci USA. 2011;108(14):5819-24.
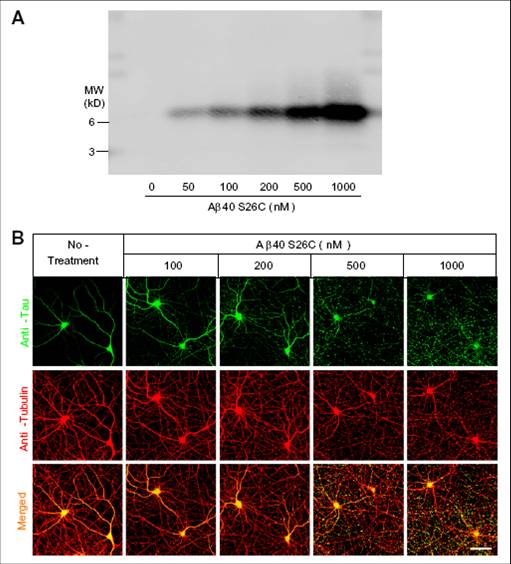
Non-fibrillar, water-soluble low-molecular weight assemblies of the amyloid β-protein (Aβ) are believed to play an important role in Alzheimer’s disease (AD). Aqueous extracts of human brain contain Aβ assemblies which migrate on SDS-PAGE and elute from size exclusion as dimers (~8 kDa) and can block long term potentiation and impair memory consolidation in the rat. Such species are detected specifically and sensitively in extracts of Alzheimer brain suggesting that SDS-stable dimers may be the basic building blocks of AD-associated synaptotoxic assemblies. Consequently, understanding the structure and properties of Aβ dimers is of great interest. In the absence of sufficient brain-derived dimer to facilitate biophysical analysis, we generated synthetic dimers designed to mimic the natural species. For this, Aβ(1-40) containing cysteine in place of serine 26 was used to produce disulphide cross-linked dimer, (AβS26C)2. Such dimers had no detectable secondary structure, produced an analytical ultracentrifugation (AUC) profile consistent for an ~8.6 kDa protein, and had no effect on hippocampal long term potentiation (LTP). However, (AβS26C)2 aggregated more rapidly than either AβS26C or wild type monomers and formed parastable β-sheet rich, thioflavin T positive, protofibril-like assemblies. Whereas wild type Aβ aggregated to form typical amyloid fibrils, the protofibril-like structures formed by (AβS26C)2 persisted for prolonged periods and potently inhibited LTP in mouse hippocampus. These data support the idea that Aβ dimers may stabilize the formation of fibril intermediates by a process distinct from that available to Aβ monomer and that higher molecular weight pre-fibrillar assemblies are the proximate mediators of Aβ toxicity.
O'nuallain B, Freir DB, Nicoll AJ, et al. Amyloid beta-protein dimers rapidly form stable synaptotoxic protofibrils. J Neurosci. 2010;30(43):14411-9.
Alzheimer's disease (AD) is the most common age-related dementia. Unfortunately due to a lack of validated biomarkers definitive diagnosis relies on the histological demonstration of amyloid-β (Aβ) plaques and tau neurofibrillary tangles. Aβ processing is implicated in AD progression and many therapeutic strategies target various aspects of this biology. While Aβ deposition is the most prominent feature of AD, oligomeric forms of Aβ have been implicated as the toxic species inducing the neuronal dysfunction. Currently there are no methods allowing routine monitoring of levels of such species in living populations. We have used surface enhanced laser desorption ionization time of flight (SELDI-TOF) mass spectrometry incorporating antibody capture to investigate whether the cellular membrane-containing fraction of blood provides a new source of biomarkers. There are significant differences in the mass spectra profiles of AD compared with HC subjects, with significantly higher levels of Aβ monomer and dimer in the blood of AD subjects. Furthermore, levels of these species correlated with clinical markers of AD including brain Aβ burden, cognitive impairment and brain atrophy. These results indicate that fundamental biochemical events relevant to AD can be monitored in blood, and that the species detected may be useful clinical biomarkers for AD.
Villemagne VL, Perez KA, Pike KE, et al. Blood-borne amyloid-beta dimer correlates with clinical markers of Alzheimer's disease. J Neurosci. 2010;30(18):6315-22.
Alzheimer's disease constitutes a rising threat to public health. Despite extensive research in cellular and animal models, identifying the pathogenic agent present in the human brain and showing that it confers key features of Alzheimer's disease has not been achieved. We extracted soluble amyloid-beta protein (Abeta) oligomers directly from the cerebral cortex of subjects with Alzheimer's disease. The oligomers potently inhibited long-term potentiation (LTP), enhanced long-term depression (LTD) and reduced dendritic spine density in normal rodent hippocampus. Soluble Abeta from Alzheimer's disease brain also disrupted the memory of a learned behavior in normal rats. These various effects were specifically attributable to Abeta dimers. Mechanistically, metabotropic glutamate receptors were required for the LTD enhancement, and N-methyl D-aspartate receptors were required for the spine loss. Co-administering antibodies to the Abeta N-terminus prevented the LTP and LTD deficits, whereas antibodies to the midregion or C-terminus were less effective. Insoluble amyloid plaque cores from Alzheimer's disease cortex did not impair LTP unless they were first solubilized to release Abeta dimers, suggesting that plaque cores are largely inactive but sequester Abeta dimers that are synaptotoxic. We conclude that soluble Abeta oligomers extracted from Alzheimer's disease brains potently impair synapse structure and function and that dimers are the smallest synaptotoxic species.
(a) TBS extracts of AD (top panel) and control (bottom panel) brains were subjected to non-denaturing SEC. SEC fractions were lyophilized and WB’d with 2G3+21F12; molecular weights (in kDa) on left. Note that Aß monomer and dimer in the AD TBS extract is recovered from material that elutes at the end of the void volume (fractions 3/4). Control (Con) brain extracts were devoid of Aß. (b) Summary LTP data (means ± SEMs) for slices treated with SEC fractions from AD or Con TBS extracts (n=6 slices for all samples), as characterized in Fig. 3a. (c) Representative WB (2G3+21F12) of IP-SEC fractionation of AD TBS and Con TBS. TBS extracts (500 µL) were immunoprecipitated with 3D6 (3 µg/mL), eluted with sample buffer and subjected to SEC. Dimer-enriched (fractions 7–8) and monomer-enriched (fractions 10–11) IP-SEC fractions were separately pooled, as were corresponding fractions from Con TBS. (d) Summary LTP data (means ± SEMs) for slices treated with IP-SEC fractions of AD and control brain TBS extracts, as characterized in Fig. 3c (Con fractions 10–11, n=5; AD fractions 10–11, n=5; Con fractions 7–8, n=7; AD fractions 7–8, n=7). (e) Mutant Aß40-S26C forms dimers under oxidizing conditions (ox), which can be reduced to monomers by treating with ß-ME (red). Silver stain was performed with 100 ng wildtype Aß40 (wt) peptide or the mutant peptide (f) Summary LTP data (means ± SEMs) for slices treated with 5, 50, or 100 nM of either wt Aß40 (black) or oxidized Aß40-S26C (red) reveals that the oxidized Aß40-S26C dimer inhibits LTP with much greater potency (100 nM Aß40-S26C, n=4; n=5 for all other treatments). The vehicle controls (plotted at 0 nM) were 50 mM ammonium acetate (n=4) for the S26C peptide and 0.1% ammonium hydroxide (n=4) for wt peptide. Shankar GM, Li S, Mehta TH, et al. Amyloid-beta protein dimers isolated directly from Alzheimer's brains impair synaptic plasticity and memory. Nat Med. 2008;14(8):837-42.

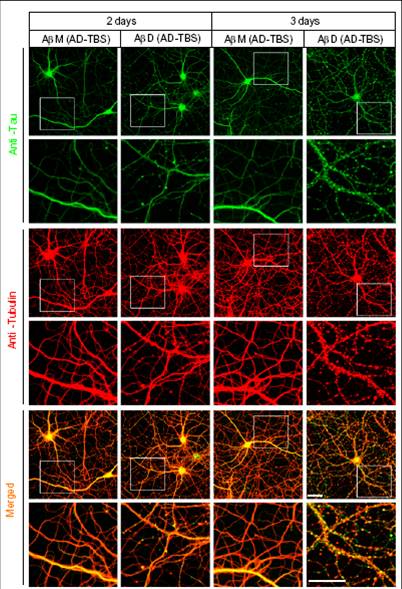 |
Higher-power confocal images showing the tau (green) and Tubulin (red) immunoreactivities of the cytoskeleton of hippocampal neurons [days in vitro (DIV) 20 or 21] after 2- or 3-d treatment with Aß monomers (M) or dimers (D) isolated by SEC from AD-TBS extracts. Regions in the white boxes are shown immediately below at higher magnification. (Scale bar, 50 µm)
|
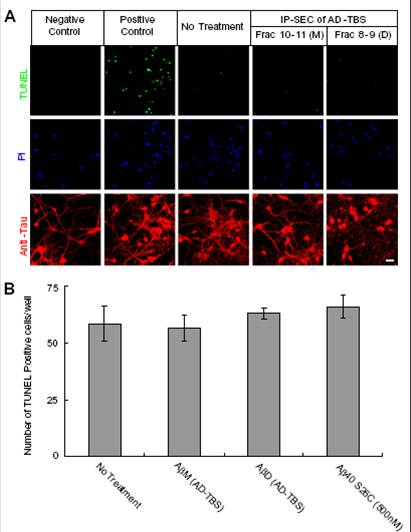 |
(A) Confocal images showing TUNEL (green), propidium iodide (PI) (blue), and the tau-reactive microtubule cytoskeleton (red) of primary hippocampal neurons (DIV21) after 3-d treatment with SEC fraction 10–11 (M, monomers) or fraction 8–9 (D, dimers) from AD-TBS. Neurons without dUTP incorporation served as negative control, and neurons treated with DNase I after fixation served as positive control. (Scale bar, 50 µm.)
(B) Histograms of the average numbers of TUNEL-positive cells in each well (~4 × 104 neurons) after the indicated treatments. Means from three independent experiments; error bars, SEM. |
| Catalog# | Product | Standard Size | Price |
|---|---|---|---|
| G-018-71 | Amyloid beta 40-S26C dimer / [A-beta S26C]2 (Human) - Purified IgG Antibody | 200 µg | $571 |
| 018-71 | Amyloid beta 40-S26C dimer/ [A-beta 40 S26C]2 (Human) | 100 µg | $382 |
| 018-92 | Amyloid beta-42 S26C dimer/ [A-beta 42 S26C]2 (Human) | 100 ug | $360 |
| 018-94 | Amyloid beta-43 S26C dimer/ [A-beta 43 S26C]2 (Human) | 100 ug | $360 |
Social Network Confirmation